Introduction to Lipids: Classification and Functions in Biology
Slides about Introduction to Lipids: Classification and Functions. The Pdf, a presentation for University students in Biology, details the main classes of lipids, their structures, and their role in determining blood groups.
See more30 Pages
Unlock the full PDF for free
Sign up to get full access to the document and start transforming it with AI.
Preview
INTRODUCTION
Lipids = a chemically heterogeneous group of compounds, characterized by water-insolubility molecules # they are highly soluble in organic solvents -> unlike other classes of biomolecules (carbohydrates, proteins and nucleic acids), lipids do not form polymers, even displaying a variety of biochemical roles,
Main classes of lipids
- Free fatty acids (Non-esterified Fatty Acids or NEFA) -> the simplest form of lipid, mainly used as a fuel -> they are characterized by a hydrocarbon chain (fatty acids = carboxylic acids with long hydrocarbon chains), that varies in length.
- Triacylglycerols -> the storage form of fatty acids.
- Phospholipids -> membrane lipids = fatty acids + a scaffold linked to a charged phosphoryl group -> amphipathic macromolecules = a polar head + nonpolar tail.
- Glycolipids -> lipids bound to carbohydrates constituting membranes.
- Steroids -> different class of lipids = polycyclic hydrocarbons -> they are precursors of hormones and the most common steroid is cholesterol (membrane structural component).
Lipid Functions
- The most common form of energy store.
- The key structural component of biological membranes.
- Enzyme cofactors, electron carriers, light- absorbing pigments, hydrophobic anchors for proteins, emulsifying agents in the digestive tract, hormones and intracellular messengers involved in signal- transduction pathways.
INTRODUCTION 7 14.007 15 30.974 N P Nitrogen [He] 2s22p3 Nonmetal Phosphorus [Ne] 3s23p3 Nonmetal 6 12.011 1 1.008 8 15.999 C Carbon [He] 2s2 2p2 Nonmetal H Hydrogen 15 Nonmetal O Oxigen [He] 2s2 2p4 Nonmetal
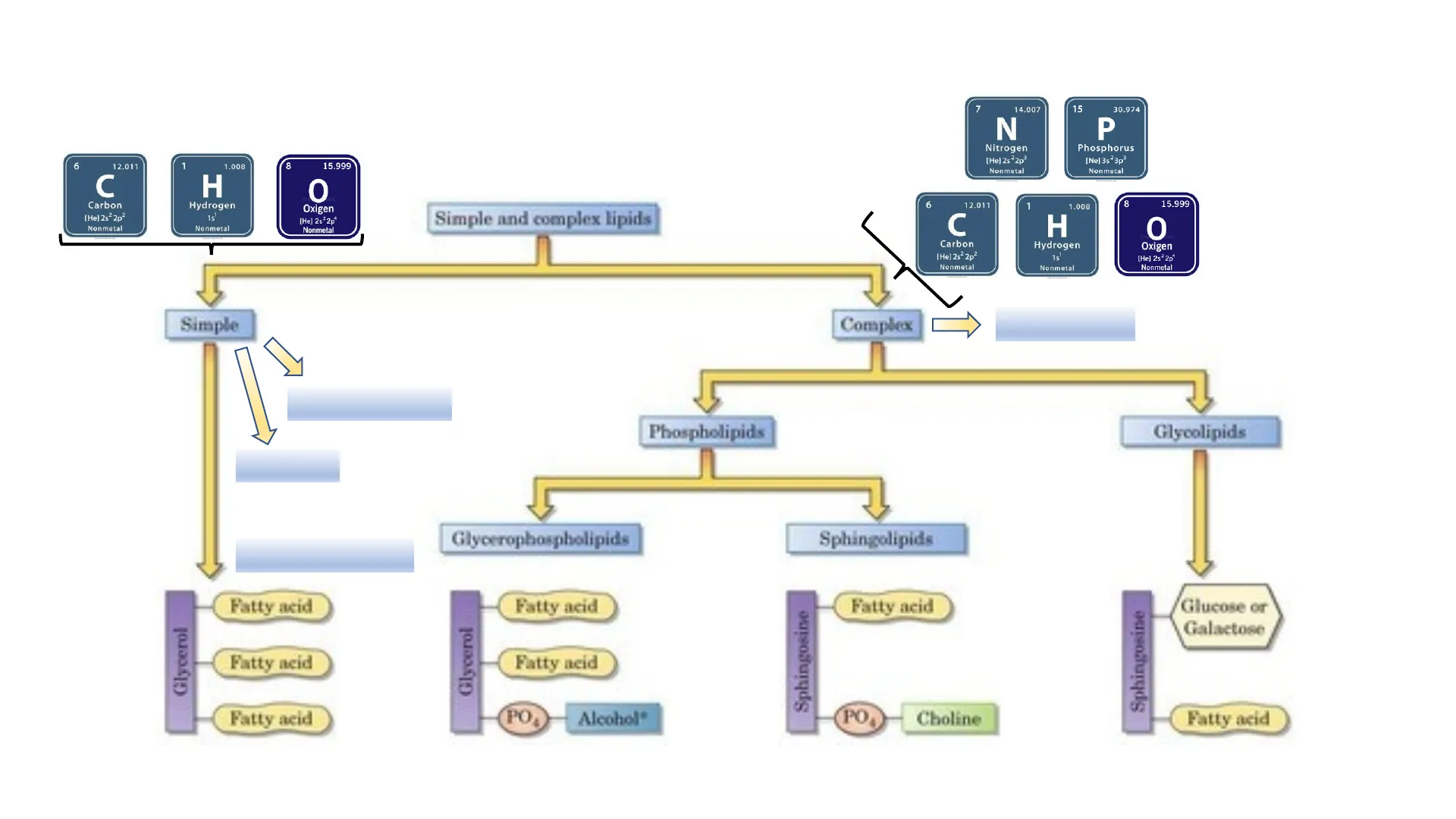
Simple and complex lipids
6 12.011 1 1.008 C Carbon [He] 2s2 2p2 Nonmetal H Hydrogen 15 Nonmetal 0 Oxigen [He] 2s22p4 Nonmetal Complex D Lipoproteins Free fatty acids Phospholipids Glycolipids Triacylglycerols Glycerophospholipids Sphingolipids Fatty acid Fatty acid Fatty acid Glycerol Fatty neid Fatty acid POD Alcohol" PO Choline Fatty acid Sphingosine Glucose or Galactose Fatty neid Sphingosine 8 15.999 Simple Steroids
FATTY ACIDS
Storage Lipids and Energy
Storage lipids = stored forms of energy -> in living organisms they are represented by fats and oils = derivatives of fatty acids, that in turn are compounds deriving from hydrocarbon derivatives -> they are characterized by high reduced state (> carbohydrates), being their complete (with CO2 and H2O final product) oxidation/combustion a highly exergonic process. Fatty acids = carboxylic acids with hydrocarbon chains ranging from 4 to 36 carbon atoms -> chains can contain single C-C bonds (fully saturated molecules) or may have one or more double bonds (unsaturated fatty acids) -> they display a double role = efficient energy fuel + building components for membrane -> carbon atoms are involved in C-C, C=C and C-H bonds # in carbohydrates there are C-C, C-H, C-O and C=O-> fats yield more energy than carbohydrates when undergoing combustion.
Fatty Acid Nomenclature
Both common and systematic names/nomenclature can be used to refer to fatty acid -> in systematic names of fatty acids, -oic acid terminology is adopted, where "oic" desinence replace final "e" of parent hydrocarbon -> 1) the C18 saturated fatty acid is familiarly known as stearic acid, but should be properly named octadecanoic acid, since octadecane represents its parent hydrocarbon with a C18 fully saturated chain # 2) oleic acid is a C18 (mono)unsaturated fatty acid with one double bond and is called octadecenoic acid # 3) linoleic acid contains two double bonds and is called octadecadienoic acid # 4) linolenic acid displays three double bonds and is named octadecatrienoic acid.FATTY ACIDS The complete systematic nomenclature of a fatty acid includes numbers, letters (Greek alphabet) and symbols, where two numbers, separated by a colon, indicate the chain length and number of double bonds -> 16:0 and 18:0 respectively denote fatty acids with no double bonds, namely palmitic acid (C16) and stearic acid (C18) # 18:1 and 18:2 refer to C18 fatty acids respectively harboring one (oleic acid) and two (linoleic acid) double bonds. Within fatty acid carbon atom chain, there is the need to distinguish the individual carbon atoms, by numbering them or labeling them with Greek alphabet letters -> carbon atoms are usually numbered starting from the carboxyl group and carbon atoms 2 and 3 can be also marked as a and B, respectively. The exact position of a double bond is indicated by the uppercase Greek alphabet letter A, followed by a superscript number -> oleic acid, harboring a double bond between Cg and C10, is properly described as 18:1(49) The terms "cis" and "trans" designate the positions of substituents (atoms of chemical group # H) on both sides of the double bond, being "cis", when substituents are on the same side, and "trans", when they are positioned on opposite sides -> cis-49-Octadecenoic acid -> this designation is used to within the full systematic name, but not to describe the carbon atom skeleton/chain
Fatty Acid Structure and Properties
A fatty acid structure can be schematically illustrated by a zigzag line, where each segment represents a single bond between two adjacent carbon atoms, while a second segment indicates the presence of a double bond. The most commonly occurring fatty acids have even numbers of carbon atoms (ranging between 12 and 24, with C16 and C18 fatty acids being the most common) and double bonds are in the cis configuration. O a 4 1 2 18 3 9 10 C 18:1(△9) cis-9-Octadecenoic acid Chemico-physical properties of the fatty acids are mainly determined by the 1) length and 2) degree of unsaturation of the hydrocarbon chain -> the nonpolar hydrocarbon chain = poor water solubility -> the longer the acyl chain and the fewer the double bonds, the lower the water solubility # the carboxylic group is polar/ionized and is responsible accounts for the (slight) water solubility of short-chain fatty acids. FFA can circulate in the blood noncovalently bound to serum albumin (= carrier protein) # they are mainly present as carboxylic acid derivatives (esters or amides), lacking the charged carboxylate group reduced water solubility and are transported through the blood primarily as lipoproteins.
Melting Point of Fatty Acids
Melting point is also affected by the length and degree of unsaturation of the hydrocarbon chain -> the longer the acyl chain, the higher the melting point (increased van der Waals interactions) + at room temperature, saturated fatty acids (C12-C24) display a waxy consistency # unsaturated fatty acids of the same lengths are oily liquids ... this is due to different degrees of packing of the fatty acid molecules. In the fully saturated compounds, molecules mostly display a fully extended form/conformation, with minimized steric hindrance -> these molecules can pack together tightly, promoting the formation of the van der Waals interaction # in unsaturated fatty acids, the presence of a cis double bonds (# trans) generates a kink in the hydrocarbon chain, thus negatively interfering with such packing attitude = weaker intermolecular interactions = marked reduction of melting points. 2 4 6 8 14 16 18 15 17 11 Octadecanoic acid (Stearic acid) mp 70℃ A saturated fatty acid cis-4º-Octadecanoic acid (Oleic acid) mp 13℃ An monounsaturated fatty acid 12 13 2 4 6 8 10 12 14 16 18 15 HO 1 3 5 7 9 11 13 15 17 16 17 18 trans-49-Octadecanoic acid (Elaidic acid) mp 46°℃ A trans fatty acid HO 1 3 5 7 9 2 4 6 8 10 HO 1 3 5 7 9 11 13 10 12 14
Fatty Acid Unsaturation and Health
Carboxyl -O group 0 -O 1 C C Hydrocarbon chain The degree and type of unsaturation in fatty acids are important factors for human health -> diet rich in high saturated or trans-unsaturated fats is correlated with high blood levels of cholesterol and cardiovascular disease (activation of inflammatory pathways in immune cells) # certain cis-polyunsaturated fatty acids -> they are essential = we cannot synthesize them. Saturated fatty acids Mixture of saturated and unsaturated fatty acids . In most monounsaturated fatty acids, the double bond is located between Cg and C10 (49) # in polyunsaturated fatty acids, the other double bonds are generally 412 and 415 · In polyunsaturated fatty acids, double bonds are neither conjugated (-CH=CH-CH=CH-) or cumulative/consecutive (-CH=CH=CH=CH-) ... but are separated by a methylene group (- CH=CH-CH2-CH=CH-).
Omega-3 Fatty Acids
Within a fatty acid chain, the carbon atoms are usually numbered starting at the carboxyl group ... but can also be numbered starting from the last/terminal carbon atom, named w and representing carbon atom number 1. 4 O 7 2 C 18 3 9 10 18:1(△9) cis-9-Octadecenoic acid a 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 20 C 7 6 5 4 3 2 1 O 20:5(45,8,11,14,17) Eicosapentaenoic acid (EPA) Those cis-polyunsaturated fatty acids (PUFAs), displaying the first a double bond between C-3 and C-4, referred to the w carbon atom, are called omega-3 (w-3) fatty acids -> they are important for human health and they are essential = they have to be introduced in human nutrition -> they are common in vegetal oils and in cold-water fish, such as salmon. w-3 fatty acids (especially those derived from marine organisms) can 1) prevent sudden death from a heart attack, 2) reduce blood triacylglycerides, 3) lower blood pressure and 4) counteract thrombosis by slightly inhibiting blood clotting. BLOOD SURE SKY HIGH 3 O 1
Omega-3 PUFAs and Diet
The "Mediterranean diet" has been associated with reduced cardiovascular risk ... it includes foods/products that were found to be rich in omega-3 PUFAs -> leafy vegetables and fish oils. These compounds includes a-linolenic acid or ALA, defined as 18:3(49,12,15), eicosapentaenoic acid or EPA, defined as 20:5(45,8,11,14,17) and docosahexaenoic acid or DHA, defined as 22:6(44,7,10,13,16,19). 0 = @-Linolenate 0 - Eicosapentaenoate (EPA) - Docosahexaenoate (DHA)
TRIACYLGLYCEROLS
Triacylglycerol Structure
Triacylglycerols = triglycerides -> the simplest form of lipids, composed of 3 fatty acids + 1 glycerol molecules, where both components are linked by an ester bond -> they can be distinguished in simple and mixed (the most naturally occurring ones) triacylglycerols based on the constituent fatty acids (the same kind of fatty acid or different fatty acid molecules in all three positions). 1CH2 3 CH2 / HO 2CH OH OH Simple triacylglycerols are named based on the constituent fatty acid -> 16:0, 18:0 and 18:1 are designed as tripalmitin, tristearin and triolein, respectively ≠ unambiguous nomenclature of mixed triacylglycerols must specify the name and position of each fatty acid. Glycerol 0 HỌC-0-C-CH2 (CH2)n-CH3 0 HC-0-C-CH2 (CH2)n -CH3 0 Three fatty acid chains HỌC-0-C-CH2 (CH2)n-CH3 Glycerol backbone Fatty acids used for energy production are stored as triacylglycerols -> triacylglycerols are generated through the attachment of three fatty acids to a glycerol molecule -> fatty acids are bond to the glycerol through ester linkages (esterification process). 1 CH2 3 CH2 O C-O 2CH 0-C =0 O C 1-Stearoyl, 2-linoleoyl, 3-palmitoyl glycerol, a mixed triacylglycerol