The Thyroid Gland: Physiology, Biosynthesis, and Hormone Actions
Document from University about The Thyroid. The Pdf details the physiology of the thyroid gland, including hormone biosynthesis, secretion, and factors influencing its function. The Pdf, suitable for University Biology students, covers oxygen consumption and thyroid hormone actions.
See more12 Pages
Unlock the full PDF for free
Sign up to get full access to the document and start transforming it with AI.
Preview
The Thyroid Gland: Physiology
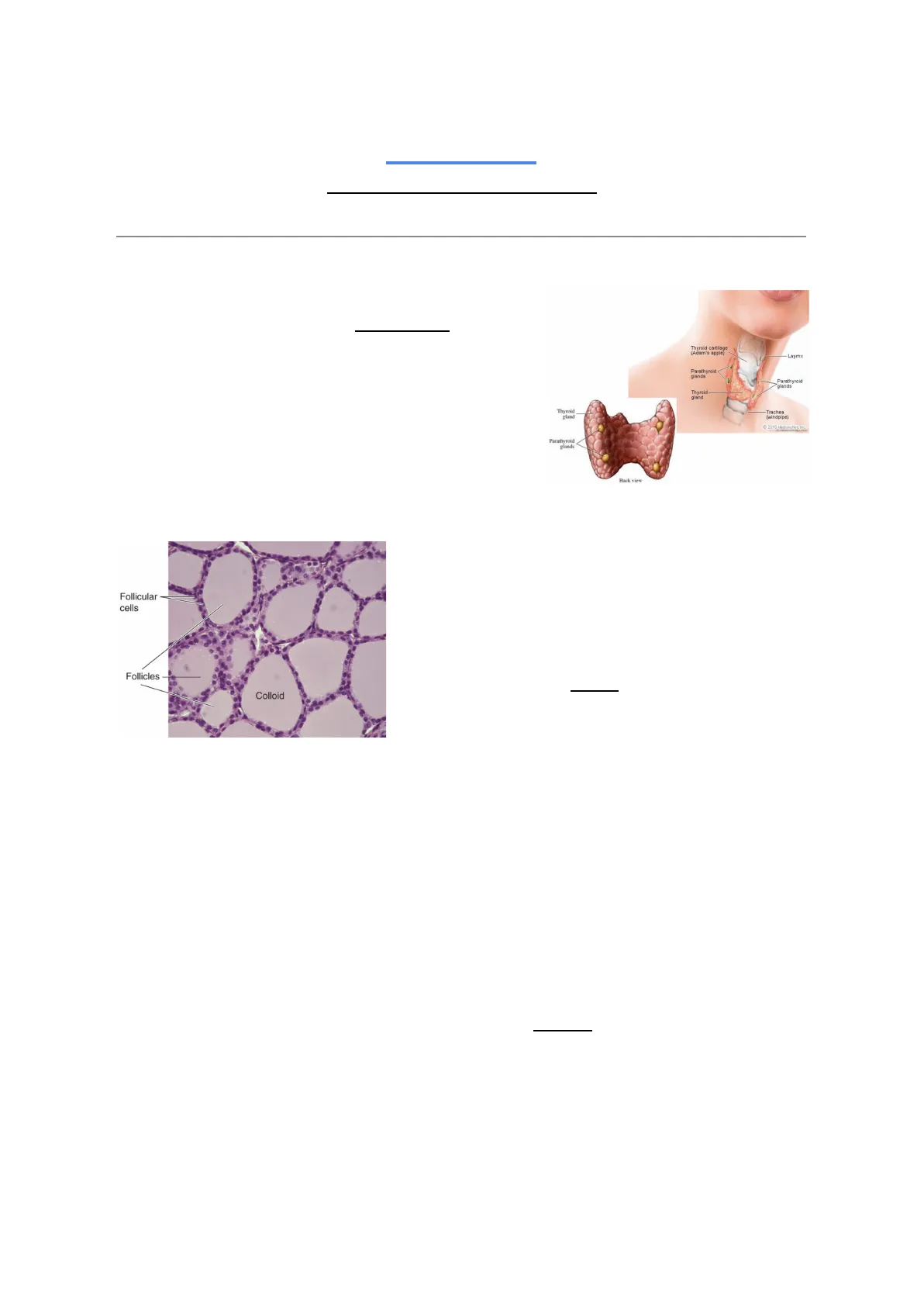
10/03/2023 The Thyroid Body At work II - physiology Couple 14 The thyroid is a gland located in the anterior part of the neck around the thyroid cartilage. Within the thyroid gland, we also have smaller glands called parathyroids, which are mostly concerned with parathormone and regulate calcium levels, reabsorption and homeostasis in general. We discussed this last semester with Professor Mazziotti Along with other hormones, the thyroid produces calcitonin, which helps the parathormone in regulating calcium homeostasis.
Thyroid Cartilage and Gland Structure
Thyroid cartilage (Adam's apple) Laymx Parathyroid lands Parathyroid lands Thyroid gland Thyroid gland Trachea (windpipe) 2010 MedicineNet, Inc. Parathyroid glands Back view Follicular cells Follicles Colloid
Thyroid Hormone Production and Storage
As regards the thyroid hormones, the thyroid gland is quite particular because most secretory glands (exocrine or endocrine) typically have granules or vacuoles within the cells where the various secretory products (hormones, enzymes etc) are contained. In this case, we have the secretory cells located around follicles, which do not contain cells but a gel - colloid (mostly constituted by high concentrations of a protein containing the thyroid hormones).
This happens mainly for two reasons:
- The hormones contain a high level of iodine (three to four iodine atoms per molecule of hormone). Iodine is not continuously present in the blood, ready to be used to form the hormones. So, the thyroid must capture iodine whenever it is around, store it and prepare the hormones depending on the availability of iodine. As a consequence, it is important that we have a large storage space.
- The huge storage cannot be performed in the usual way (by accumulating the products in a vesicle within a cell) because the thyroid hormones are very lipophilic and so they would exit the vesicle and exert their actions.
The thyroid hormones are not actually trapped after being synthesized, but they are already trapped before synthesis. The hormones are based on the amino acid tyrosine and they are synthesized within a protein that contains a lot of tyrosine residues - so, not synthesized starting from free tyrosine. When you want to release the hormone, you digest the protein and release it.
Thyroid Gland Regulation
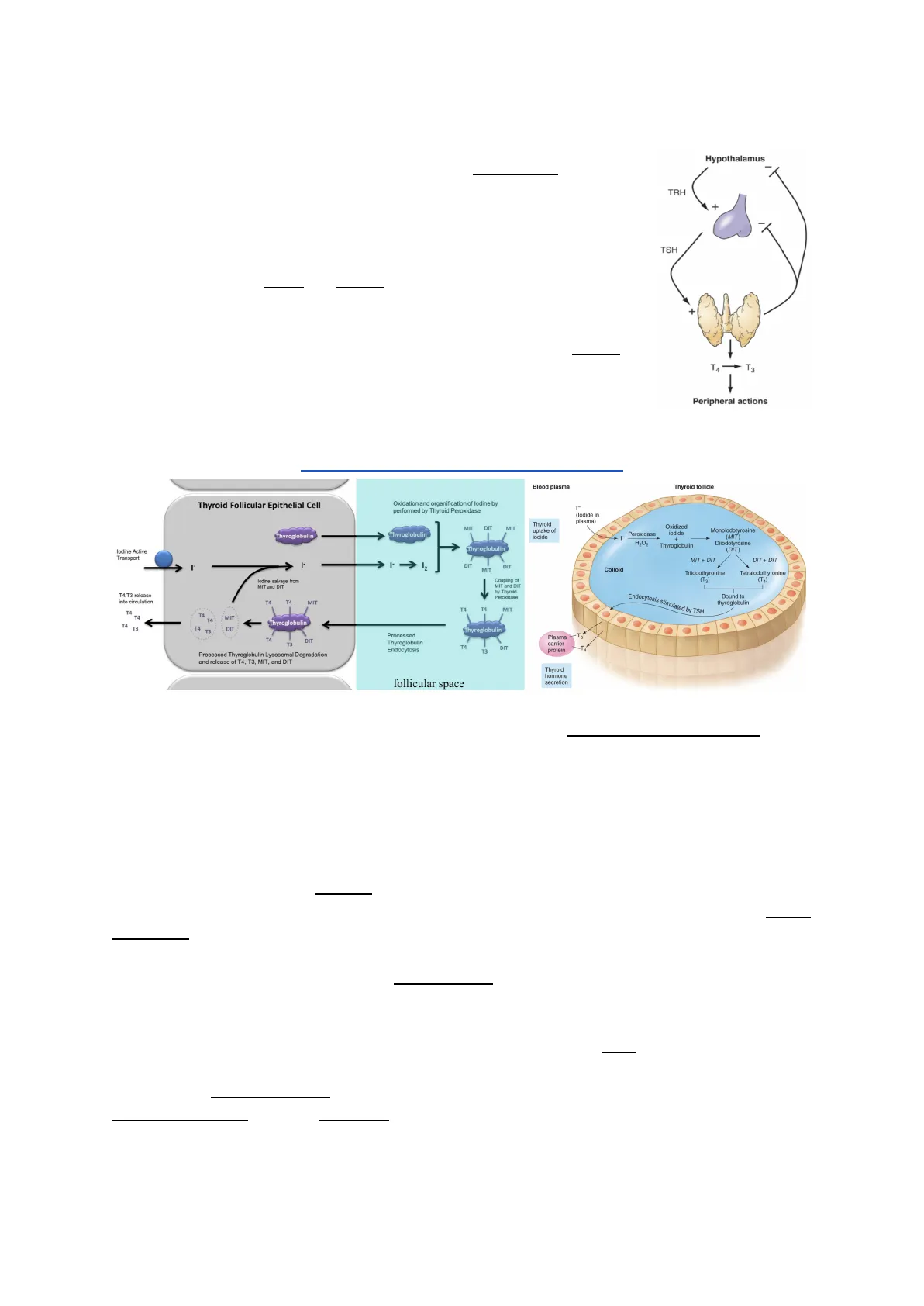
1The activity of the gland is under the control of thyrotropin (or TSH - thyroid stimulating hormone). TSH is released by thyrotrophs or cells in the anterior pituitary gland that respond to TRH (Thyrotropin-releasing hormone), produced by the hypothalamus. The action of TSH is tropic and trophic: it is directed to stimulate the release of the hormone, but it is also a trophic action on the gland. If the gland cannot produce the hormone and the TRH + TSH levels grow because of that, the gland will proliferate and increase in size (this leads to the Goiter).
Hormone Biosynthesis and Secretion
Hypothalamus TRH + TSH + T4 T3 Peripheral actions BIOSYNTHESIS AND SECRETION Thyroid follicle Thyroid Follicular Epithelial Cell Oxidation and organification of lodine by performed by Thyroid Peroxidase (lodide in plasma) MIT DIT MIT Thyroid uptake of lodide - 1 Oxidized lodide + Thyroglobulin Monolodotyrosine (MIT Diiodotyrosine (DIT) DIT + DIT - DIT DIT Colloid Triiodothyronine (Ta) Tetraiodothyronine (TA) lodine salvage from MIT and DIT Coupling of MIT and DIT by Thyroid Percoidase Endocytosis stimulated by TSH T4 MIT Thyroglobulin 14 13 T4 DIT Thyroglobulin T4 DI T3 Processed Thyroglobulin Endocytosis T4 DI T3 Plasma carrier protein Processed Thyroglobulin Lysosomal Degradation and release of T4, T3, MIT, and DIT Thyroid hormone secretion follicular space
Iodine Uptake and Organification
The main problem with the thyroid is getting enough iodine to synthesize the hormone when it is needed. The iodide is effectively captured from the blood by the sodium-iodide transporter (active transport), which uses the driving force of sodium to transport in a combined way one ion of sodium and one ion of iodide. NB. Iodide = ion Iodine = element Once it is in the cell, it will be handled by another transporter, which is a transporter of iodide in exchange of chloride, called pendrin - often used to exchange ions in a relatively unspecific way (ex. Iodide for Bicarbonate). Pendrin will transfer the iodide in the follicle, where we have iodide peroxidase. This enzyme will oxidize iodide into iodine and attach it to the phenyl group of tyrosine. In this way, the phenyl group becomes iodinated and this happens, as we said before, not on free tyrosine, but in a specific protein called thyroglobulin. The cell produces thyroglobulin and sends it to the follicle where it constitutes the gel. In this way, thyroglobulin will contain some mono-iodinated tyrosines (MIT) and some tyrosines with two iodine molecules attached (DIT). The fusion between one MIT and one DIT will lead to the production of triiodothyronine (T3), while the fusion between two DIT will lead to the production of tetraiodothyronine (T4 or thyroxine). Thyronine is the fusion of two tyrosines (also called oxophenylalanine).
2 T4 TA MIT T4 T4 MIT T4 T4 T4 Bound to thyroglobulin T4/T3 release into circulation Thyroglobulin lodine Active Transport Thyroglobulin Thyroglobulin Peroxidase H-O2 MIT + DIT MIT Blood plasma T3I I I H- I NH2 0 HO- 0 CH2-CH-C HO- -CH-CH-C OH OH I I I Triiodothyronine (T3) Fesce said to revise the basic amino acids Glycine is the simplest amino acid, alanine is a 3-carbon amino acid, oxophenylalanine is tyrosine. In these molecules, we have a tyrosine attached to another tyrosine. One of them is attached to the chain and we can see this by the fact that it has the COOH and NH2. The second tyrosine is cut in such a way that it doesn't have the amino acid portion and it attaches with a bond to the tyrosine still in the sequence of the protein. Thyronine has two phenyl groups and 3 or 4 iodines: these molecules make thyronine much more lipophilic than expected. Being an amino acid, it should be hydrophilic but, since it has phenyl groups and iodines (both really lipophilic), it becomes lipophilic itself. When you release this molecule, it is very lipophilic and it needs proteins to travel in the blood. Lipophilic substances need these proteins because usually proteins have an hydrophilic surface and whatever is lipophilic is usually contained inside the protein. The protein can also have a pouch or a fold where there is a lipophilic environment to host lipophilic substances in order for them to not form lipid droplets in the blood.
Thyroid Hormone Activity and Deiodinase Enzymes
Depending on where you attach the iodine, T3 is not always the same:
- If you attach the two iodines to the tyrosine that maintains its amino acid structure (the one inside the chain of the protein), you have the correct form called T3
- If you attach the second iodine on the phenyl group not part of that tyrosine, then you have a reverse T3. This reverse T3 is not active and does not have hormonal activity
In the blood, we have both T3 and T4, but 80% is T4, which is more lipophilic. T4 is essentially the hormone that more easily enters the cells. When it enters the cells, however, it doesn't have any transcriptional activity and doesn't bind to the receptors with a significant affinity. Still, it can be cleaved inside the cells into T3 (the active hormone) or into the reverse T3 (inactivating the T4). Depending on which deiodinase enzyme is activated and expressed, each cell can decide how strongly it will respond to the thyroid hormone:
- If it expresses the DI01, T4 becomes T3
- If it expresses the DI03, T4 becomes the reverse T3 because DI03 cleaves the wrong iodine
1 I I I Fetus HO- CH2CH-COOH 1 > HO- -CH2CH-COOH Inactivation I I NH2 I NH2 3,3',5'-Triiodothyronine (reverse T3, rT3) Peripheral Target Tissues 1 I > HO- CH2CH-COOH Activation - NH2 I 3,5,3'-Triiodothyronine (T3) 3 1 O= Thyroxine, or tetraiodothyronine (T4) NH2 .0 3,5,3',5'-Tetraiodothyronine (thyroxine, T4)>99% of circulating T3 & T4 are protein-bound, primarily to Thyroid-Binding-Globulin. Peripheral Tissues 15% TA Other degradative pathways 80% 45% D3 - T4 40%| D1 Inactive reverse T3 PTU 1 T3 . T3 Biological activity D3 Thyroid Inactive Ta It is a way for the different cells to be more or less responsive. It is not a self-controlling mechanism, which instead occurs on the hypothalamus, pituitary gland and thyroid itself (so, not on the target cells). In many cases, cells may try to down-regulate their response to a stimulus, but Fesce doesn't think this is a case of self-control/down-regulation. It is interesting that these deiodinase enzymes exist because they can be inhibited by specific drugs: for example, Propylthiouracil (PTU) is able to inhibit IOD1. If you inhibit IOD1, you reduce the responsiveness of the cells (T4 is transformed mainly into reverse T3). Situations of hyperthyroidism (excessive concentration and production of the hormones) can be treated pharmacologically in this way (within certain limits). The exact sequence of the biochemical changes was discussed with Professor Greco. The important concept to remember is that the secretory cells in the thyroid have a kind of activity and organization, which is different from the other endocrine systems. They keep producing thyroglobulin proteins and secreting them into the follicles. The final synthesis of the hormone actually occurs in the acellular compartment - here we can find the enzymes, but no cytosol. When the cell has to release the hormones, it engulfs a portion of the follicular content by endocytosis. Then, this content is processed in order to digest the protein and release the hormones. Along with the hormones, the cell will also release some DIT and MIT still present in the protein and not yet combined to form a complete and mature hormone (they are sent back in the gel of the follicle to be possibly used for the formation of other hormones). The production and the release of the enzymes follow two different paths. Essentially, the cells synthesize thyroglobulin and send it to the follicles. Then, when they are asked to release the hormones, they take a part of the follicular content, digest the protein and release the hormone. This is the reason why, in response to TSH and TRH, you cannot separate the two aspects. Even if the resulting hormone is ineffective because of a lack of iodine, the stimulus will continue and will keep producing thyroglobulin filling the follicle (this is why it becomes so big, the gland proliferates and we have the goiter). There is a third form of deiodinase, which is DI02, mostly expressed in the hypothalamus and in the pituitary gland. It has the function of transforming T4 into T3. These cells are very sensitive to the presence of the hormone and, as a consequence, they can generate a proper negative feedback control. The hypothalamus and the pituitary gland need to monitor the level of the thyroid hormones: either you generate a transduction system that is able to sense the concentration of T4 or you provide 4 20%