Proteins: Classification, Primary and Secondary Structures
Slides from University about Proteins. The Pdf, a presentation for University Biology students, details protein classification, primary structure with examples like hormones and sickle cell anemia, and secondary structure including alpha helix and beta sheet.
See more14 Pages
Unlock the full PDF for free
Sign up to get full access to the document and start transforming it with AI.
Preview
PROTEIN CLASSIFICATION
Proteins are biopolymers formed by many amino acids (more than 80) joined together by peptide bonds.
Based on their chemical composition, proteins are divided into two main categories:
- simple proteins, if they are formed only by amino acids;
- conjugated proteins, if they are made up of amino acids and a prosthetic group (a molecule of a non-protein nature) which can be a lipid, a glycide, a nucleic acid or a metal group.
-5-
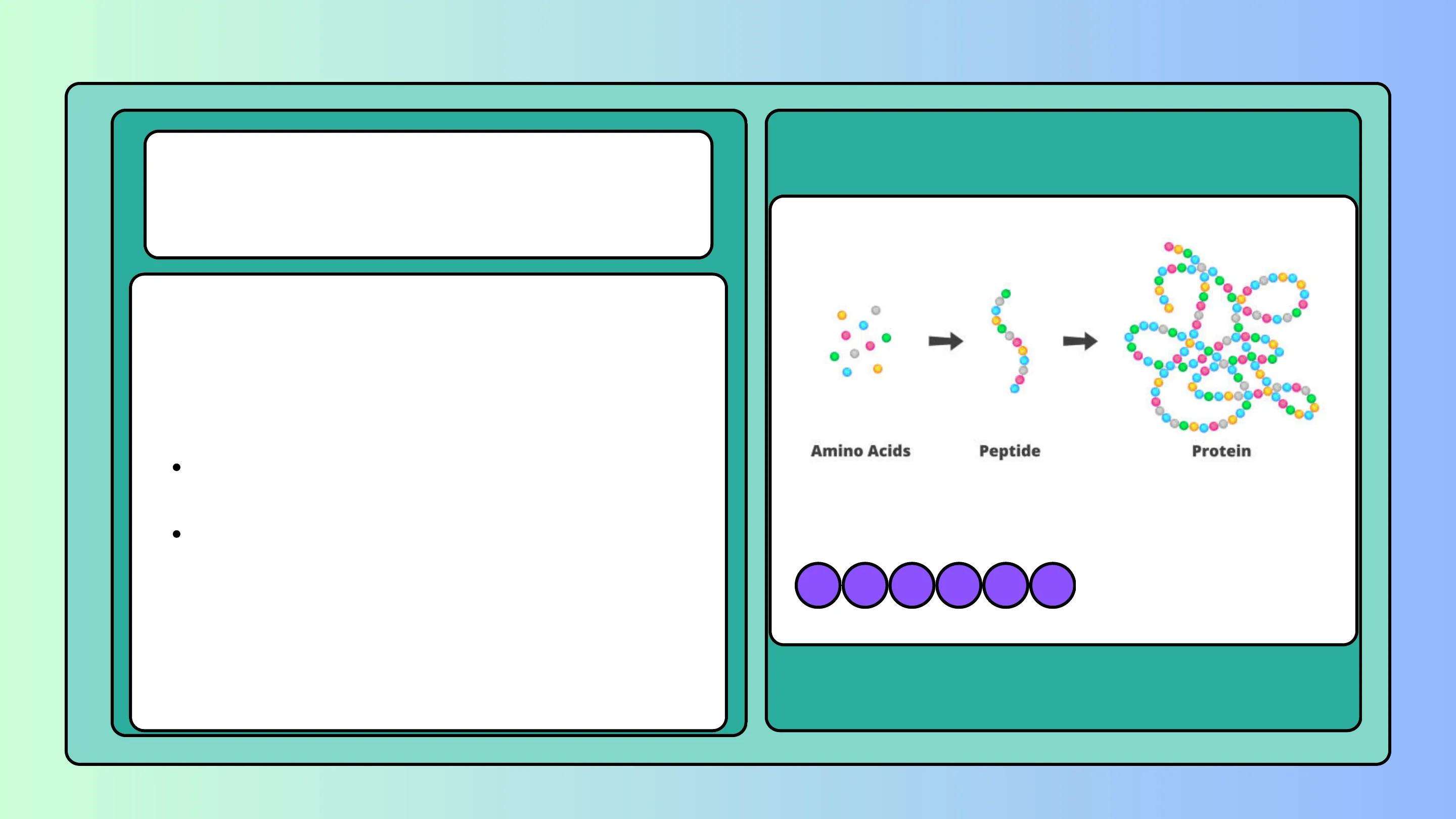
Amino Acids Peptide Protein
Protein FUNCTIONS
The infinite possibility of combining amino acids in different sequences gives rise to proteins with different functional roles in living systems.
Depending on their biological function, proteins can be divided into the following classes:
- structural proteins, which form tissues and organs; keratin (present in nails and hair), collagen and silk fibroin belong to this group.
- catalytic proteins, such as enzymes, which regulate chemical reactions in cells;
- contractile and movement proteins, which allow the contraction of muscle cells and the movement of cilia and flagella (tubulin);
- transport proteins, such as hemoglobin, which helps transport oxygen in the blood;
elica - alfa cheratina
R R
Hemoglobin a Chain B Chain Heme group Oxygen molecule iStock® Credit: Rujirat Boonyongron ₿ Chain a Chain 1387416895
FUNCTIONS
- reserve proteins, such as ferritin, which accumulates iron ions Fe2 in the liver, or ovalbumin, which functions as a reserve substance in the embryo;
- defense proteins, such as antibodies, which protect the body from pathogens;
- regulatory proteins, such as some hormones produced by endocrine glands, which regulate various metabolic processes in organisms.
ovoalbumin
SHAPE
Based on the shape they take in space, proteins can be divided into fibrous and globular.
- Fibrous proteins are formed by two or three polypeptide chains arranged next to each other and linked together by disulfide bonds, as in the case of keratin, or by hydrogen bonds (as in collagen), to form a single filament.
- Globular proteins are formed by polypeptide chains folded on themselves in compact structures, of a more or less spherical shape, determined by numerous intramolecular and ionic interactions. Globular proteins include enzymes, hormones, transport proteins (such as blood hemoglobin), reserve proteins and defense proteins (antibodies).
Fibrous Protein Globular Protein
CHEMICAL STRUCTURE OF PROTEINS
By examining a protein in its spatial configuration, it is possible to identify four levels of organization which are called primary, secondary, tertiary and quaternary structure.
The Primary Structure of a Protein is its Amino Acid Sequence
The primary structure is defined by the sequence of amino acids linked with peptide bonds in the polypeptide chain. Each protein has its own specific sequence and its biological function depends on it.
Here, for example, are the first 20 amino acids (out of a total of 124) in the protein ribonuclease from a cow:
KETAAAKFERQHMDSSTSAA
Primary structure Secondary structure Tertiary structure Quaternary structure
Amino acid a-Helixes Polypeptide chains Complex of protein molecule
PRIMARY STRUCTURE
For example, two protein hormones, oxytocin and vasopressin, differ only in two amino acids but perform completely different biological functions:
- oxytocin is responsible for the contraction of the uterine muscles before and during childbirth;
- vasopressin promotes the reabsorption of water at the renal level.
Primary structure Amino acids Oxytocin Vasopressin NH2 Pro Lely NH2 Pro)Arg Cys Cys Gly Cys)Cys CONH2 CONH. Tyr Asn Tyr Asn Ile GIn Phe Gln Gly
PRIMARY STRUCTURE
The replacement, elimination or addition of an amino acid in the amino acid sequence (due to a gene mutation) can modify the biological activity of a protein and be the basis of serious pathologies. In hemoglobin, the protein that binds and transports oxygen in the blood, a genetic mutation can cause red blood cells to take on a particular sickle shape, responsible for sickle cell anemia.
The primary structure determines the subsequent levels of structural organization and the final shape of the protein, on which its function depends. In fact, the side chains of the amino acids favor or hinder the realization of a certain shape in the space of the polypeptide chain.
NORMAL REO BLOOD CELL SICKLE REO BLOOD CELL
SECONDARY STRUCTURE
A protein's secondary structure consists of regular, repeated spatial patterns in different regions of a polypeptide chain.
There are two basic types of secondary structure, both determined by hydrogen bonding between the amino acids that make up the primary structure: the a helix and the ß pleated sheet.
Many proteins contain regions of both a helix and B pleated sheet in the same polypeptide chain.
HYDROGEN BONDS ALPHA HELIX BETA - PLEATED SHEET
ALPHA HELIX
The a (alpha) helix is a right-handed coil that turns in the same direction as a standard wood screw. The R groups extend outward from the peptide backbone of the helix.
The coiling results from hydrogen bonds that form between the 8+ hydrogen of the N_H of one amino acid and the 8- oxygen of the C=O of another. When this pattern of hydrogen bonding is established repeatedly over a segment of the protein, it stabilizes the coil.
C- C I-Z H ON~C-C. - O C C. I N O / HO H O N O=O1 H C H C C. 1 - N N O O Alpha helix C N . N - C C -C/
BETA HELIX
A B (beta) pleated sheet is formed from two or more polypeptide chains that are almost completely extended and aligned. The sheet is stabilized by hydrogen bonds between the N_H groups on one chain and the C=O groups on the other. A B pleated sheet may form between separate polypeptide chains or between different regions of a single polypeptide chain that is bent back on itself.
O O H H O=0 H C-C-NIC_ C_N C-C-NICON -C-N.C.CAN Nao H H H H . .. . . O C C C H O C 1=0 Z-I 0=0 I-Z 0=0-0 Z-I =0-0 Z-I H 0=0 =0-0 Z-I I -Z C 1 I-Z 1. IN 1 0 Beta pleated sheet H -C C-N.
TERTIARY STRUCTURE
The tertiary structure is defined by the shape that the protein assumes after being stabilized by hydrogen bonds, disulfide bonds, ionic interactions and van der Waals interactions that form between amino acids.
In the tertiary structure, the hydrophobic side chains are positioned inside the structure, while the hydrophilic ones, given their solubility in water, are positioned outside.
A protein is able to carry out its specific biological activity only when it has assumed its definitive three-dimensional structure (protein folding).
Hydrophobic interactions CH CH2 OI ... 0=0- H3C H3C CH3 CH3 Polypeptide backbone CH HO-C Hydrogen bond CH2 -CH2 -S-S -- CH2 Disulfide bridge 0 -- CH2 -NH3+ 0-C-CH2 lonic bond
QUATERNARY STRUCTURE
The quaternary structure is defined by the association of two or more polypeptide chains (subunits) and is stabilized by hydrogen bonds, interactions between apolar R groups and disulfide bonds.
Hemoglobin, the oxygen transport protein in red blood cells, is an example of a globular protein with a quaternary structure: it is made up of four polypeptide chains, two a and two ß; each subunit is linked to a prosthetic group (heme), each containing an iron ion Fe2+.
Antibodies, the defense proteins produced by B lymphocytes, are also made up of four polypeptide chains (two light and two heavy) linked together by disulfide bonds.
B1 B2
PROTEIN DENATURATION
The chemical bonds responsible for the secondary, tertiary and quaternary structure of a protein are weak bonds. Consequently, high temperatures (which increase the kinetic energy), extreme pH values (which change the dissociation of the amino and carboxyl groups) and organic solvents (which change the arrangement of the hydrophilic and hydrophobic groups) can lead to the breaking of these bonds.
DENATURATION Folded Protein Unfolded Protein
The breaking of the chemical bonds that stabilize the secondary, tertiary and quaternary structure leads to the denaturation of the protein, that is, to the loss of the structure and, therefore, of the function of the protein.
When a protein, by denaturing, forms new intramolecular and intermolecular bonds, the denaturation process is irreversible.