Organización del citoplasma: citoesqueleto, filamentos de actina y microtúbulos
Diapositivas sobre la organización del citoplasma: Citoesqueleto. El Pdf, de nivel universitario, explora la estructura y función de los filamentos de actina, microtúbulos y filamentos intermedios en biología, con ilustraciones detalladas.
Ver más35 páginas

Visualiza gratis el PDF completo
Regístrate para acceder al documento completo y transformarlo con la IA.
Vista previa
Organización del Citoplasma
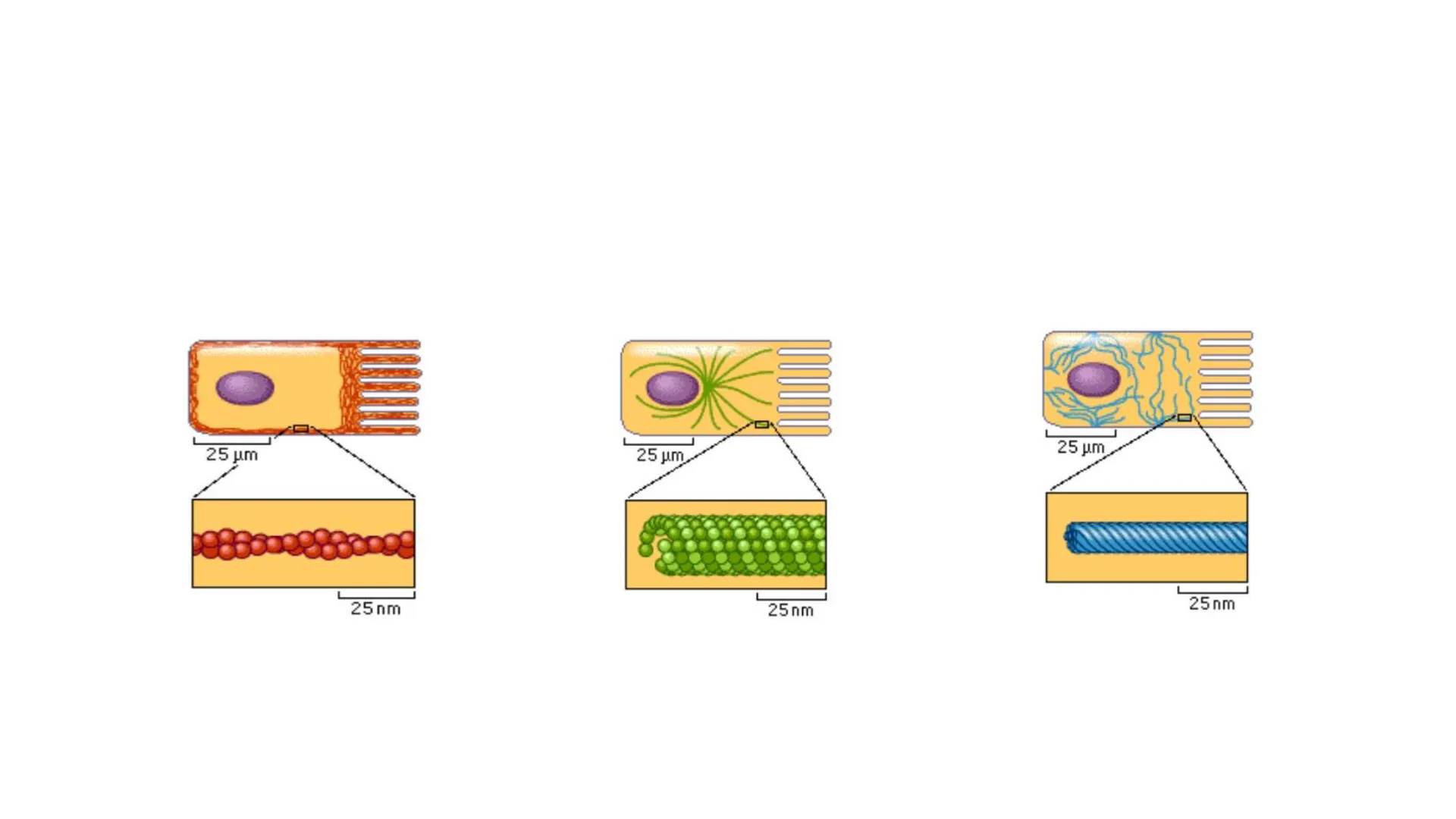
Organización del citoplasma: Citoesqueleto Carolina Baez Fonaudiología Bases de las ciencias biológicas en la salud 2024EN EL CITOPALSMA ENCONTRAMOS AL CITOESQUELETOY SE COMPONE DE 3 TIPOS DE FILAMENTOS
- FILAMENTOS DE ACTINA
- MICROTUBULOS
- FILAMENTOS INTERMEDIOS
0 25 μm 25 um 25 nm 25 nm 25 um 25 nm 8-9 nm 24 nm 10 nmShear 1 TensionEL CITESQUELETO PARTICIPA EN LAS UNIONES CELULARES APICAL
- microvillus terminal web of actin adherens junction desmosome nucleus + + +
- hemidesmosome + basal lamina BASAL intermediate filaments microtubules actin microfilaments
Flexibilidad de los Filamentos del Citoesqueleto
- En muchas células eucariontes se encuentran 3 tipos de filamentos de citoesqueleto, los cuales son fundamentales para la organización espacial de estas células.
- Filamentos intermedios: proveen fuerza mecánica y resistencia a la deformación.
- Microtúbulos: determinan la posición de los organelas y transporte directo intracelular.
- Filamentos de actina: determinan la forma de las células y son necesarios para el movimiento celular.
- deformation - microtubules intermediate filaments actin filaments deforming force Molecular Biology of the Cell. 4th edition. Alberts B, Johnson A, Lewis J, et al. New York: Garland Science; 2002.FILEMNTOS ACTINA 100 nm 25 nm actin molecule plus end plus end NH2 T HOOC 37 nm ATP (ADP when in filament) minus end minus end (B) (C) 25 nm (A)
Ubicación Celular de Filamentos de Actina
Ubicación en la célula de FILAMENTOS DE ACTINA C (A) (B) (C) (D) Figure 17-28 Essential Cell Biology 3/e (@ Garland Science 2010) A-Microvellosidades B-Haces contractiles C-Protusiones de una célula en movimiento D-Anillo contractili)
Cortex crosslinked networks ii) iv) iii) Stress fibers antiparallel contractile structures Lamellipodium FilopodiumOlson, M. F., & Sahai, E. (2008). The actin cytoskeleton in cancer cell motility. Clinical & Experimental Metastasis, 26(4), 273-287. stress fibres pseudopodium ** filopodia lamellipodium
Polimerización y Despolimerización del Filamento de Actina
POLIMERIZACIÓN Y DESPOLIMERIZACION DEL FILAMENTO DE ACTINA P Hydrolysis of ATP DO AND TADP ADP OO ADPODP ADP ADP ATP ATPOORATP Plus end ATP ADP ADP ADECADE ATP ATP ATP Exchange of ATP for ADP ADP d P barbed end pointed end nd Actin-ADP-Pi TADPOO ADP ATP Actin-ATP Actin-ADP
Procesos de Polimerización de Filamentos de Actina
POLIMERIZACIÓN DE LOS FILAMENTOS DE ACTINA nucleation (lag phase) elongation (growth phase) steady state (equilibrium phase)
- Pasa por 3 procesos
- NUCLEACIÓN:
- ELONGACIÓN:
- EQUILIBRIO: La polimerización de subunidades de actina pura en filamentos ocurre después de una fase de retraso. 100 concentration of monomers at steady state = C: % actin subunits in filaments actin filament with subunits coming on and off growing actin filament actin subunits 0 oligomers (A) time after salt addition
Fases de Polimerización de Actina
POLIMERIZACIÓN DE LOS FILAMENTOS DE ACTINA nucleation (lag phase) elongation (growth phase) steady state (equilibrium phase) elongation (growth phase) steady state (equilibrium phase) 100 concentration of monomers at steady state = C _ % actin subunits in filaments actin filament with subunits coming on and off actin filament with subunits coming on and off growing actin filament actin subunits 0 oligomers (A) time after salt addition 100 Ce unchanged by addition of nuclei % actin subunits in filaments growing actin filament 0 preformed filament seeds added here (B) time after salt addition
Nucleación de Filamentos de Actina
NUCLEACIÓN:
- Un polímero helicoidal se estabiliza mediante múltiples contactos entre subunidades adyacentes. En el caso de la actina, dos moléculas de actina se unen relativamente debilmente entre sí, pero la adición de un tercer monomero de actina para formar un trímero hace que todo el grupo sea más estable. La adición adicional de monómeros puede tener lugar sobre este trímero, que por lo tanto actúa como un núcleo para la polimerización
Rol de la Profilina en la Elongación de Filamentos de Actina
LA PROFILINA DESEMPEÑA UN PAPEL EN LA ELONGACIÓN DE LOS F.A. REGULANDO DE LA POLIMERIZACIÓN DE LA ACTINA. Box 1 | The engine: core actin polymerization machinery Profilin-℮ a Spontaneous Barbed end Nucleation Elongation T Pointed end F-actin G-actin F-actin nucleus Capping protein Su función principal es unirse a los monomeros de actina globular (G-actina) y facilitar su incorporación en los extremos de crecimiento de los filamentos de actina (F-actina), lo que promueve la elongación de estos filamentos.
Función del Complejo ARP2/3 en Filamentos de Actina
ARP2/3 ES UN COMPLEJO PROTEICO QUE DESEMPEÑA UN PAPEL CRUCIAL EN LA NUCLEACIÓN Y RAMIFICACIÓN DE LOS FILAMENTOS DE ACTINA EN LAS CÉLULAS EUCARIOTAS. b b Facilitated Formin Enhanced elongation daughter filament Branching nucleation Arp 2/3 ENA/VASP Profilin- ARP2/3 complex (inactive) mother filament 70° Este complejo tiene la capacidad única de inducir el crecimiento de nuevos filamentos de actina a partir de los ya existentes en un ángulo de aproximadamente 70 grados, lo que resulta en la formación de una estructura de red ramificada.
Asociación de Filamentos de Actina con Contactos Focales
LOS FILAMENTOS DE ACTINA SE ASOCIAN CON LOS CONTACTOS FOCALES Molecular architecture of FAs Actin st-060 för00 Myosin Myosin Myosin Zyn Vincuin Vro a-Action Focal contact proteins FAK FAK Pasión Yp1300130 Plasma imembrane boo Intogrins Extracelular matrix NRM&CB 6:56, 2005 Composition of a focal adhesion
- Actin filament a-actinin Paxillin Vinculin Focal adhesion kinase Tensin Talin Integrin \ Integrin Sro Extracellular Matrix
Actina y Desplazamiento Celular
ACTINA Y DESPLAZAMIENTO CELULAR actin cortex lamellipodium substratum cortex under tension actin polymerization at plus end protrudes lamellipodium PROTRUSION movement of unpolymerized actin myosin II contraction ATTACHMENT AND TRACTION focal contacts (contain integrins) Figure 16-86 Molecular Biology of the Cell 5/e (@ Garland Science 2008) La célula extiende protrusiones en su frente o extremo directo
- Modelo de movimiento celular mediado por polimerización de actina Las protrusiones se adhieren a la superficie por donde la célula se desplaza gracias a los contactos focales El resto de la célula es arrastrado mediante tracción hacia los puntos de anclaje
Microtúbulos y Polimerización de Tubulina
MICROTÚBULOS B-tubulin GTP tubulin heterodimer (= microtubule subunit) protofilament lumen plus end 1 50 nm minus end (B) a-tubulin (C) microtubule Polimerización de tubulina GTP GTP @ Tubulin ₿ Tubulin GTP cap (+) end 1 1 GDP micro- tubule (-) end = = Protofilament assembly 2 Sheet assembly Microtubule elongation
Inestabilidad Dinámica de Microtúbulos
LA POLIMERIZACION INESTABILID DINÁMICA Y DESPOLIMERIZACION GENERA GTP-tubulin dimer B GTP exchangeable GTP GTP GTP GTP (-) (+) straight protofilament Preexisting microtubule GTP HYDROLYSIS CHANGES SUBUNIT CONFORMA AND WEAKENS BOND IN THE POLYMER GDP GDP GDP curved protofilament DEPOLYMERIZATION GDP GDP GDP-tubulin dimer GDP 1 GDP-GTP EXCHANGE GTP (B) High concentration of GTP-bound free tubulin Low concentration of GTP-bound free tubulin 1 accidental loss of GTP cap CATASTROPHE rapid shrinkage 1 regain of GTP cap RESCUE rapid growth with GTP-capped end etc. sólo se añade tubulina GTP (la tubulina GDP tiende a despolimerizar) Addition of GTP-bound tubulin less stable region of microtubule containing GDP-tubulin dimers GTP cap rapid growth with GTP-capped end GROWING SHRINKING (C) Tubulin with bound GDP Tubulin with bound GTP GTP cap Stable Unstable GDP cap (A)
Proteínas en Polimerización y Despolimerización de Microtúbulos
HAY PROTEINAS QUE PARTICIPAN EN LA POLIMERIZACION Y DESPOLIMERIZACION DEL MICROTÚBULO. catastrophe factor (kinesin-13) DESTABILIZATION frequency of catastrophes increased GTP cap on plus end of microtubule STABILIZATION XMAP215 frequency of catastrophes suppressed and growth rate enhanced
Microtúbulos en Centrosomas
ENCONTRAMOS MICROTUBULOS EN LOS CENTROSOMAS nucleating sites (y-tubulin ring complexes) + + + + pericentriolar material + + + + + + + pair of centrioles + + + + + + microtubules growing from 7-tubulin ring complexes of the centrosome (B) + + + x O D (C) (A)
Composición de Centriolos
LOS CENTRIOLOS SE COMPONEN DE MICROTUBULOS (A) distal appendages mother centriole pericentriolar material daughter centriole (C) O A B C microtubule triplet
Estructura de Cilios
Cilios inner sheath L radial spoke central singlet microtubule nexin inner dynein arm outer dynein arm t 1 A microtubule B microtubule- plasma membrane 100 nm outer doublet microtubule(A) myosin-I 70 nm y y (B) myosin-I t vesicle (C) + myosin-I Figure 17-36 Essential Cell Biology 3/e (@ Garland Science 2010) plasma membrane(A) myosin-II molecule head tail 1 150 nm myosin heads (B) myosin-II filament bare region 1 pm Figure 17-38 Essential Cell Biology 3/e (@ Garland Science 2010) + myosin-Il + Figure 17-39 Essential Cell Biology 3/e (@ Garland Science 2010) plasma membrane
Contracción Muscular
CONTRACCIÓN MUSCULAR
- El sarcómero se acorta producto del desplazamiento de los filamentos de actina por sobre los de miosina-II sin que ocurra acortamiento de ninguno de ellos. sarcomere thick filament (myosin filament) thin filament (actin filament) Z disc Z disc (A) CONTRACTION RELAXATION (B) Figure 17-42 Essential Cell Biology 3/e (@ Garland Science 2010)
Filamentos Intermedios
FILAMENTOS INTERMEDIOS NH2 COOH a-helical region of monomer NH2 COOH (B) coiled-coil dimer NH2 COOH 48 nm NH2 COOH COOH NH2 NH2 COOH NH2 staggered tetramer of two coiled-coil dimers lateral association of 8 tetramers (D) (E) addition of 8 tetramers to growing filament (A) COOH
Unión de Filamentos Intermedios a Hemidesmosomas
LOS FILAMENTOS INTERMEDIOS SE ENCUENTRAN UNIDOS A LOS HEMIDESMOSOMAS basal cell of epidermis basal lamina defective keratin filament network hemidesmosomes (C)
Tipos de Filamentos Intermedios
INTERMEDIATE FILAMENTS CYTOPLASMIC NUCLEAR keratins vimentin and vimentin-related neurofilaments nuclear lamins in epithelia in connective tissue, muscle cells, and neuroglial cells in nerve cells in all animal cells
Epidermólisis Bullosa Simplex
Epidermolysis Bullosa Simplex mutations in keratin 14 (K14) or K5, the type I and II intermediate filament (IF Epidermis Dermis Hypodermis Basal layer Simplex Junctional Dystrophic ECM a K14 null c K14 null d WT 7 epi epi Bilater Blister b WT hf hf Chf Dermis Dermis Coulombe, P. A., & Lee, C .- H. (2012). Defining keratin protein function in skin epithelia: epidermolysis bullosa simplex and its aftermath. Journal of Investigative Dermatology, 132(3 Pt 2), 763-775.
Filamentos Intermedios Nucleares
Filamentos intermedios nucleares CYTOSOL nuclear envelope nuclear pore nuclear lamina NUCLEUS OČ chromatin Se encuentran por la cara interna de la envoltura nuclear
Filamentos Intermedios en Uniones Celulares
Filamentos intermedios en uniones celulares
- Desmosomas: unen célula-célula
- Hemidesmosomas: unen célula-matriz extracelular Desmosomas 1 epithelial tissue Hemidesmosomas basal lamina 2 connective tissue collagen fibers