Gas Transport in the Human Body: Oxygen and Carbon Dioxide Exchange
Slides from Department of Medicine & Surgery about Gas Transport. The Pdf, a university-level Biology document, details the transport of oxygen and carbon dioxide, including the oxygen-hemoglobin dissociation curve and factors influencing it.
See more36 Pages
Unlock the full PDF for free
Sign up to get full access to the document and start transforming it with AI.
Preview
PHYSIOLOGY LECTURES. 2ND YEAR OF MEDICINE.
2024/2025 DR ANA CHECA -ROS MD, PHD DEPARTMENT OF MEDICINE & SURGERYSTRUCTURE
Oxygen Transport
Oxygen Transport from Alveoli to Capillaries
· From Alveoli to Capillaries: · As per the physical principles of gas diffusion, O2 diffuses from the alveoli to the capillaries because pO2 is higher in the former (according to a pressure gradient):
Alveolus Air in and out
CO 0 2 O 2 - Blood vessel (capillaries)
Oxygen Transport in Blood
· Transport in Blood: · Partial pressure of the O2 pumped out to the aorta is around 95 mmHg red = oxygen rich blue = oxygen poor pO2=95 mmHg mOxygen Transport · Transport in Blood: . 97% of 02 in blood is transported from the lungs to the tissues by the haemoglobin (Hb) · The haemoglobin is the main protein in the red blood cells: it is composed of 4 subunits, each of them may carry one O2 molecule
HEMOGLOBIN (Hb) * CAN BIND UP TO 4 O2 MOLECULES ONE PER SUBUNIT >250,000,000 Hb x4 Oz/Hb = >1,000,000,000 O2 Each red blood cell contains millions of Hb molecules, meaning that each red blood cell may transport up to billions of O2 molecules Extracted from: www.osmosis.orgOxygen Transport · Transport in Blood: · In a healthy adult, Hb concentration in blood is 15 grams of Hb/100ml of blood · As 1 gram of Hb transports up to 1.34 ml of O2, this means that around 20 ml of O2 (15 x 1.34) are transported by Hb in 100 ml of blood
BOUND to HEMOGLOBIN ML O2 20.1 100 ml blood Extracted from: www.osmosis.orgOxygen Transport · Transport in Blood: · The binding of O2 to Hb is reversible and depends on the amount of O2 dissolved in blood . Therefore, we say that Hb saturation with O2 (amount of O2 bound to Hb in blood) is directly proportional to the partial pressure of O2
25 100 MMHg MMHg V 0% OXO2 25% 1×02 50% 2×02 75% 3×02 100% 4x 02 Extracted from: www.osmosis.orgThe Oxygen-Haemoglobin Dissociation Curve shows the relationship between Hb saturation with O2 (%) and the pO2 in blood (mmHg)
100 Hemoglobin saturation (%) 90 Oxygenated blood leaving the lungs 80 70 60 50 Reduced blood returning 40 from tissues 30 20 10 0 0 10 20 30 40 50 60 70 80 90 100 110 120 130 140 Pressure of oxygen in blood (Po2) (mm Hg) Extracted from: Guyton & Hall PhysiologyThe Oxygen-Haemoglobin Dissociation Curve shows the relationship between Hb saturation with O2 (%) and the pO2 in blood (mmHg)
At a pO2= 95mmHg (pO2 in arterial blood), Hb saturation is 97%
100 Hemoglobin saturation (%) 90 Oxygenjated blood leaving the lungs 80 70 At a pO2= 40mmHg (pO2 in venous blood), Hb saturation is 75% 60 50 40 Reduced blood returning from tissues 30 20 10 0 0 10 20 30 40 50 60 70 80 90 100 110 120 130 140 Pressure of oxygen in blood (Po2) (mm Hg) Extracted from: Guyton & Hall Physiology
Factors Influencing Oxygen-Haemoglobin Affinity
· Transport in Blood: · There are three factors that influence the affinity of Hb for O2 and change the oxygen- haemoglobin dissociation curve:
- CO2 concentration
- Body temperature
- 2,3-DPG concentrationOxygen Transport · Transport in Blood: · There are three factors that influence the affinity of Hb for O2 and change the oxygen- haemoglobin dissociation curve:
- CO2 concentration: · As the blood passes through the tissues, CO2 is released from the tissues to the blood · CO2 reacts with water in blood, forming carbonic acid (H2CO3), which splits into bicarbonate ion (HCO3-) and hydrogen ion (H+)
CARBONIC ANHYDRASE CO2 + H2O H,CO 2 3 HCO3- + H+ CARBONIC ACID BICARBONATE ION HYDROGEN ION OSMOSIS. Extracted from: www.osmosis.orgOxygen Transport · Transport in Blood: · There are three factors that influence the affinity of Hb for O2 and change the oxygen- haemoglobin dissociation curve:
- CO2 concentration: · The hydrogen ions and CO2 bind to Hb, decreasing the affinity of Hb for O2 · This decrease in affinity forces the Hb to unload O2 into the tissues (shift of the oxygen- haemoglobin dissociation curve to the right)
Factors influencing the Oxygen-Haemoglobin Dissociation Curve
100 - 90 80 70 60 Shift to the right means less affinity of Hb for O2 50 Shift to right: 30 - (1) Increased hydrogen ions (2) Increased CO2 - 20 (3) Increased temperature 10 (4) Increased BPG 0 0 10 20 30 40 50 60 70 80 90 100 110 120 130 140 Pressure of oxygen in blood (Po2) (mm Hg) Extracted from: Guyton & Hall Physiology Hemoglobin saturation (%) 40Oxygen Transport · Transport in Blood: . There are three factors that influence the affinity of Hb for O2 and change the oxygen-haemoglobin dissociation curve:
- CO2 concentration: · The effect of CO2 on the oxygen-haemoglobin dissociation curve is called Bohr effect, and it allows the release of O2 from blood into the tissues as they release CO2 to blood
100- % SATURATION OF HAEMOGLOBIN LOW PCO 80- · HIGH PCO2 60- 40- 20- 0- 0 2 4 6 8 10 12 14 PARTIAL PRESSURE OF OXYGEN (KPa) Extracted from: www.osmosis.orgOxygen Transport · Transport in Blood: · There are three factors that influence the affinity of Hb for O2 and change the oxygen- haemoglobin dissociation curve:
- Body temperature: · Increased body temperature reduces the strength of the chemical bond between Hb and O2, shifting the curve to the right
TEMPERATURE - AFFECTS the STRENGTH of the BOND BETWEEN O, & HEMOGLOBIN Extracted from: www.osmosis.orgFactors influencing the Oxygen-Haemoglobin Dissociation Curve
100 - 90 80 70 60 Shift to right means less affinity of Hb for O2 50 Shift to right: 40 30 - (1) Increased hydrogen ions (2) Increased CO2 20 (3) Increased temperature - 10 (4) Increased BPG 0 0 10 20 30 40 50 60 70 80 90 100 110 120 130 140 Pressure of oxygen in blood (Po2) (mm Hg) Extracted from: Guyton & Hall Physiology Hemoglobin saturation (%)Oxygen Transport · Transport in Blood: · There are three factors that influence the affinity of Hb for O2 and change the oxygen- haemoglobin dissociation curve:
- Body temperature: · The effect of temperature on the curve is a defence mechanism during exercise: during physical activity, body temperature increases, shifting the curve to the right and facilitating the release of O2 from the Hb to the muscles, which require more O2 Extracted from: www.osmosis.orgOxygen Transport · Transport in Blood: . There are three factors that influence the affinity of Hb for O2 and change the oxygen-haemoglobin dissociation curve:
- 2,3-DPG (2,3-diphosphoglyceric acid) · 2,3-DPG is produced in situations of hypoxia (low O2), due to high altitude, or vascular problems · 2,3-DPG reduces affinity of Hb for O2, shifting the curve to the right and forcing the unload of O2 to tissues
HYPOXIC CONDITIONS ->2,3 DPG PRODUCTION Extracted from: www.osmosis.orgFactors influencing the Oxygen-Haemoglobin Dissociation Curve
100 - 90 80 70 60 Shift to right means less affinity of Hb for O2 50 Shift to right: 40 30 - (1) Increased hydrogen ions (2) Increased CO2 20 (3) Increased temperature 10 (4) Increased BPG - 0 0 10 20 30 40 50 60 70 80 90 100 110 120 130 140 Pressure of oxygen in blood (Po2) (mm Hg) Extracted from: Guyton & Hall Physiology Hemoglobin saturation (%)Oxygen Transport · Transport in Blood: . There are three factors that influence the affinity of Hb for O2 and change the oxygen- haemoglobin dissociation curve:
- 2,3-DPG (2,3-diphosphoglyceric acid) · The 2,3-DPG is a defence mechanism of our body against hypoxia, as it facilitates the release of O2 from Hb to the tissues
HYPOXIC CONDITIONS ->2,3 DPG PRODUCTION 1 Extracted from: www.osmosis.org
Clinical Implications of Oxygen-Haemoglobin Dissociation Curve
· Transport in Blood: · Clinical Implications of the Oxygen-Haemoglobin Dissociation Curve: · CO is a gas that binds Hb at the same site as O2 does, and with 250 times more affinity As you can see of the figure, Hb saturation is 97% with only a concentration of CO=0.4 mmHg (in comparison with 95mmHg of O2)
100 90 Hemoglobin saturation (%) 80 70 - 60 With only 0.4 mmHg of CO, Hb saturation is 97% 50 40 30 20 10 0 . 0 0.1 0.2 0.3 0.4 Gas pressure of carbon monoxide (mm Hg) Extracted from: Guyton & Hall PhysiologyOxygen Transport · Transport in Blood: · Clinical Implications of the Oxygen-Haemoglobin Dissociation Curve: · CO poisoning: . It is produced by inhalation from fires, combustion fumes . Even if only a small amount of CO is inhaled, it is able to displace O2 from the Hb binding site and make the Hb unable to carry O2 · The main symptoms are neurological (confusion, dizziness), as the brain is not receiving enough O2
What Is Carbon Monoxide Poisoning? ( 1) It's caused by excessive exposure to carbon monoxide, which builds up in the bloodstream 6 00 Symptoms include nausea, chest pain, and irregular heartbeats, but unconsciousness and death can occur EMT It's commonly treated with pressurized oxygen to clear the blood verywell It's most easily prevented with a carbon monoxide alarm Extracted from: Guyton & Hall Physiology
Oxygen Transport from Capillaries to Tissues
· From Capillaries to Tissues: · 02 diffuses from the capillaries (pO2=95mmHg) to the tissue cells according to a pressure gradient, as pO2 in the interstitial space surrounding the tissues is 40mmHg · 02 diffusion to the tissues stops when the pO2 in the capillaries descends until being the same as in the interstitial space (40mmHg)
Arterial end of capillary 40 mm Hg Venous end of capillary Po2 = 95 mm Hg Tissue Po2 = 40 mm Hg O Extracted from: Guyton & Hall Physiology
CO2 Transport
CO2 Transport from Tissues to Capillaries
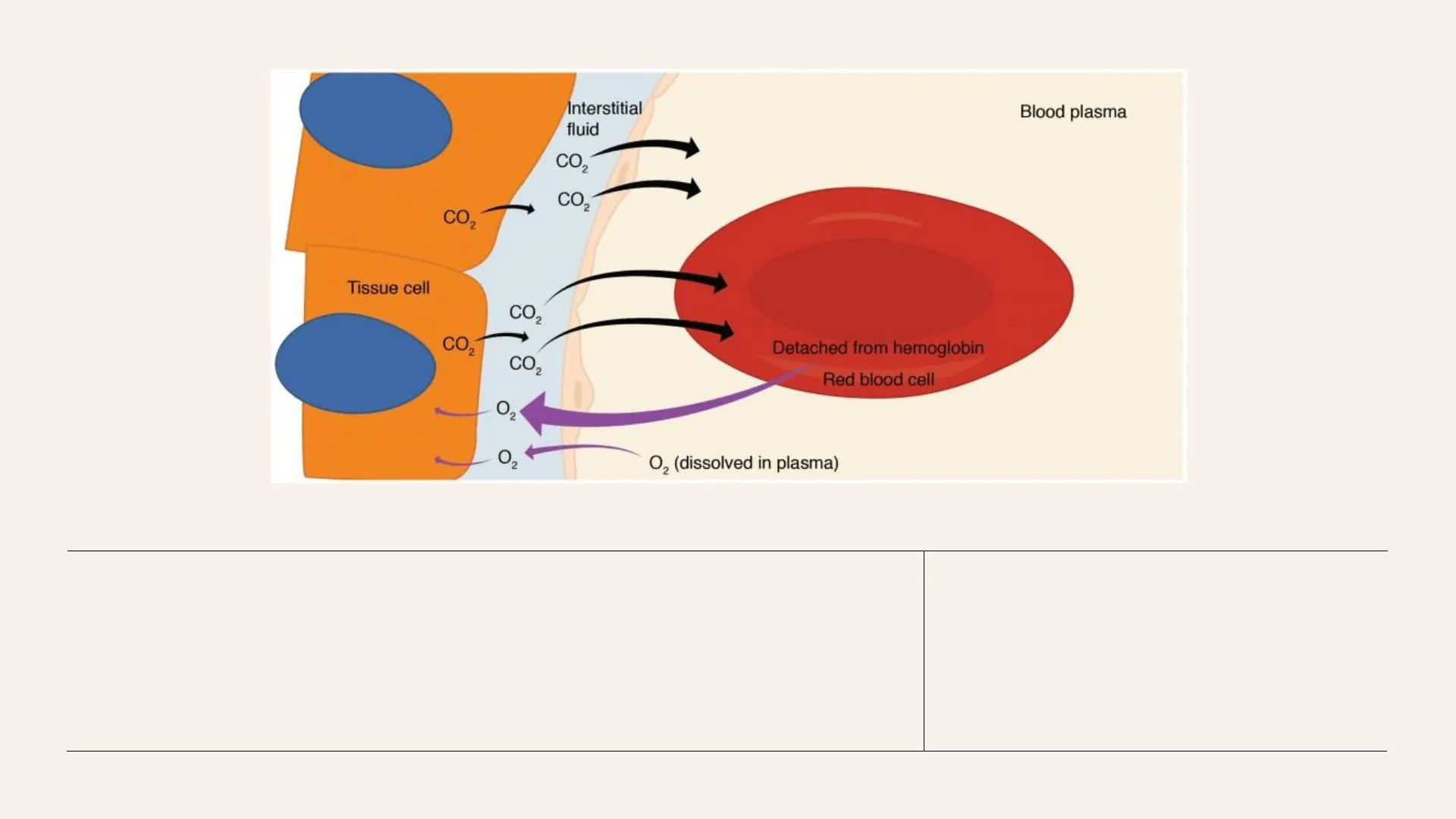
· From Tissues to Capillaries: · CO2 diffuses from the tissues to the capillaries according to a pressure gradient: pCO2 in the cells is 45 mmHg, whereas pCO2 in arterial blood is 40 mmHg · Although the pressure gradient required for CO2 diffusion is far less than the one required for O2, CO2 diffuses because its solubility is 20 times higher than in the case of O2
Arterial end of capillary Venous end of capillary 45 mm Hg Pco2 = 40 mm Hg Tissue Pco2 = 45 mm Hg O Extracted from: Guyton & Hall Physiology