Natural Products and Secondary Metabolites in Pharmaceutical Biotechnology
Document from Alessia Galli about Natural Products. The Pdf, a detailed set of notes for University Biology students, explores natural products and secondary metabolites, including their production, antibiotic resistance, clavulanic acid, and glycopeptides.
See more56 Pages

Unlock the full PDF for free
Sign up to get full access to the document and start transforming it with AI.
Preview
NATURAL PRODUCTS
Natural products are fundamental for the development of the pharmaceutical industry. They can be divided into 3 categories:
- Primary metabolites: amino acids, nucleotides, vitamin B12, organic acids (citric acid)
> Secondary metabolites: antibiotics, cyclosporin A, statins, Taxol, bio-pesticides
- Others: industrial enzymes and recombinant DNA products
Secondary metabolites are often called specialized metabolites and are typically produced by microbes or plants. The microbial producers are usually: Actinomycetes (48%), Ascomycota (34%) and other bacteria (18%) like Bacillus, Pseudomonas, Myxobacteria, Cyanobacteria. Microbes can produce many valuable products, such as:
- Biomass > yeast and probiotics
- Products from anaerobic metabolism > ethanol
- Products from incomplete oxidations > citric acid
- Products from secondary metabolism > antibiotics
- Enzymes, polysaccharides, and heterologous proteins
WHAT ARE SECONDARY METABOLITES?
The definition was introduced by a plant physiologist in 1961 (Bu'Lock): secondary metabolites are not essential for cell life and are not found in every growing cell. They're not produced equally by all the cells, but by specialized organisms. These metabolites are produced only under certain conditions and only by certain microorganisms. They are low molecular weight molecules biosynthesized from one or more general metabolites by a wider variety of pathways than those involved in general metabolism. Secondary metabolites derive from primary ones and because of this they have a higher molecular weight, but a lower molecular weight compared to usual polymers. Some microbes, like actinomycetes1 and ascomycota2, are usually exploited to produce secondary metabolites. Each strain can produce different kinds of metabolites. These two groups are historically known as the most prolific groups. The primary metabolism is needed to grow and reproduce, but secondary metabolism is needed to interact with the environment. This specialization is always consequent of a morphological differentiation and not all microbes have developed a secondary metabolism. E. coli doesn't have specific morphological differentiation and it doesn't produce secondary metabolites; this is because it lives in a very rich environment (gut). Actinomycetes is a differentiating prokaryotic organism, they get their name from fungi because they're able to produce mycelium. They produce hyphae very similar to fungi's hyphae, substrate mycelium + aerial mycelium, at one point they also produce spores. The ecological meaning of this differentiation is colonization and to exploit the environment. They live in soil, with very low nutrients so they need to colonize the soil as much as they can. When the nutrients end, they develop an aerial mycelium to continue living. The mycelium's cells then turn into spores, and they're carried away to grow somewhere else. This behavior is very similar to plants. They need to differentiate to survive. The biochemical differentiation is correlated with the way the microbes live, so it is important to replicate the right environment to make them produce the secondary metabolites.
HOW ARE THE SECONDARY METABOLITES PRODUCED?
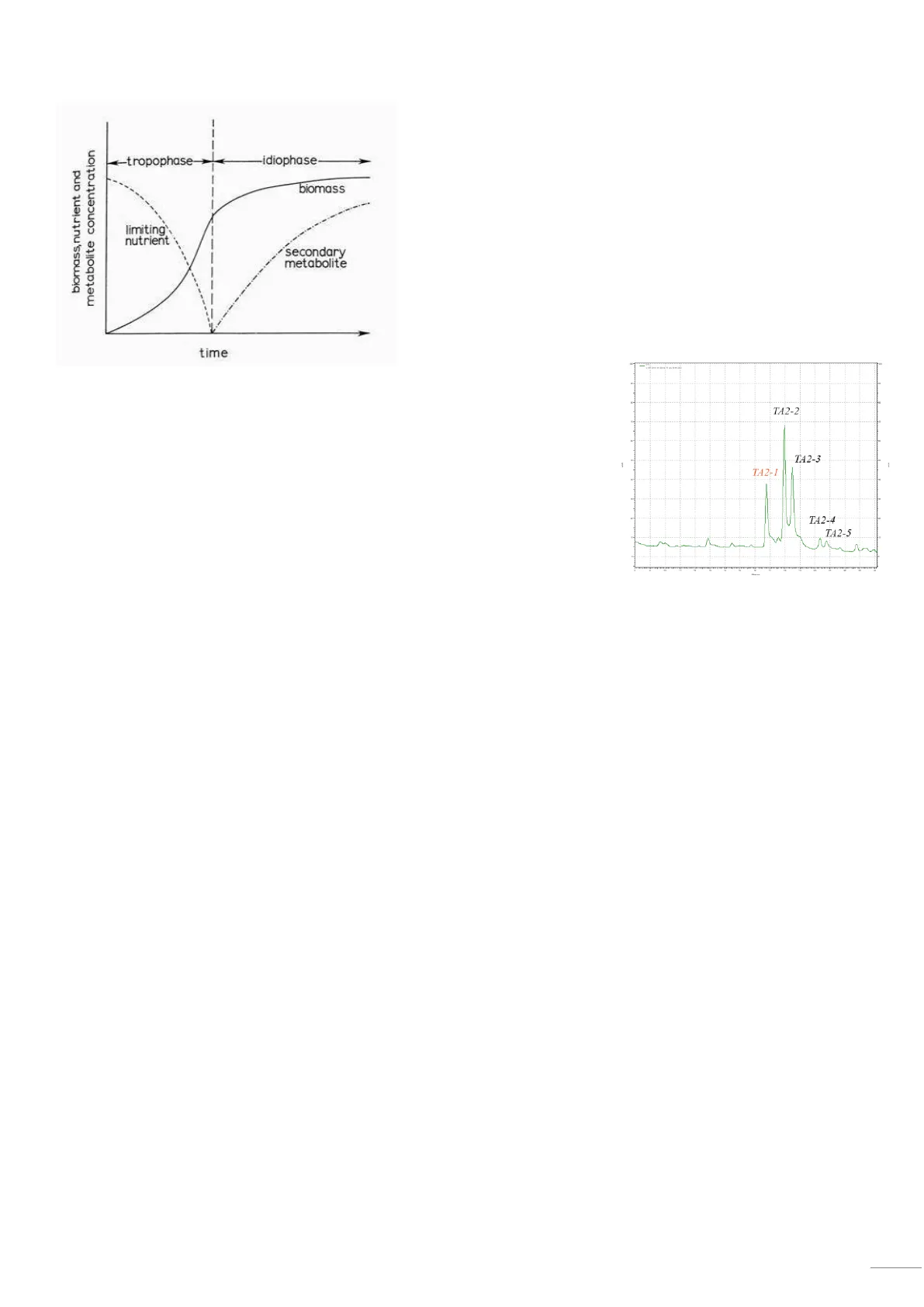
biomass, nutrient and metabolite concentration 4-tropophase- -idiophase- biomass limiting' nutrient secondary metabolite time Secondary metabolites are produced during the stationary phase, while primary metabolites are produced during the growing phase. If you handle your liquid cultures in a batch mode3 (closed system), you will have a stationary phase of growth where secondary metabolites can be produced. Most of the secondary metabolites are produced as a group of closely related structures (complex structures). In a HPLC analysis we can find different peaks, which means we have slightly different molecules. HPLC profile coupled with a UV detector of a fermentation sample. The molecules can be seen because they have double bonds and aromatic structures. You can also use HPLC coupled with mass spectroscopy. Cultivation part > where you can analyze when the production happens and how much the microbes produce. The next step would be to purify the molecules produced (most expensive step). TA2-2 TA2-3 TA2-1 TA2-4 TA2-5
WHAT ARE SPECIALIZED METABOLITES?
Secondary metabolites (1961): plant and microbial metabolites not essential for cell life and not found in every growing cell. (Bu'Lock) Small bioactive molecules (2000s): low-molecular weight organic compounds with an extraordinary diversity of molecular structures and activities produced by living organisms. (Julian Davis) Specialized metabolites (2012): small bioactive molecules made by defined, specialized and regulated biosynthetic pathways and involved in highly specific interactions with cellular targets. (Dubrovnik Summer School) These molecules are still called natural products in the industry, but they also include plant products. It's extremely important for the secondary metabolites to have highly specific targets so that they don't end up attacking our organism. For example: penicillin targets the cell wall and in particular the cross-linking bonds in the peptidoglycan, these structures are not present in the human cells. Chemists can produce many different molecules, but we then need to check if they're active in some ways. Secondary metabolites are for sure already active on something, because otherwise the cells wouldn't be producing them.
ANTIBIOTICS
They are the first specialized metabolites discovered thanks to Fleming (penicillin) and Waksman (streptomycin). They both won the Nobel prize for the discovery. Streptomycin is not used anymore to cure TBC because it had severe side effects (like deafness). The first definition of antibiotics was: A chemical substance of microbial origin that possess antimicrobial activities. They then changed it to: Low molecular weight chemical substances produced by microorganisms, which at a low concentration inhibit the growth of other microorganisms. Waksman was working in a big University using a systematical approach, now known as Waksman platform. This is a biological activity-guided screening:
- Systemically collect, enrich, and isolate soil microorganisms (actinomycetes and fungi)
He developed methods to isolate these microorganisms from the soil
o o He focused on soil because it is very rich with these microorganisms
- Growing them in axenic4 cultures
- Testing the culture broths for their ability to inhibit the growth of pathogens
o He took drops of the extracellular medium and put them on a confluent plate, to see if there was an inhibition halo Recovering the active substances produced by the specific strains
- The Waksman platform has been used by everyone in the industry. It was revolutionary because it went against the method of the chemists. Chemistry groups were interested in finding lots of new molecules that could be used as drugs, but their approach was to first describe the molecule structures and try to understand if the beneficial effects of the plant of origin could be attributed the extracted molecules. For them, studying the biological activity was the last step. Waksman did the exact opposite: the goal was to find a biological activity and then focus on understanding the chemistry of the molecules involved later. For example: they discovered actinomycin, a very powerful anti-microbial, but it was then discovered that it was also active on mammalian cells and so caused several side effects. It was discarded as an anti-microbial, but it has now been repurposed as an anti-tumoral drug.
HIGH-THROUGHPUT SCREENING
Waksman process was the base for the further development, and it made scientists interested in taking the process and make it more industrial by introducing the concept of high-throughput cultivation and screening. Scientists started to collect hundreds of samples of soils and from other environments (like oceans). Because of this, they needed to use many different isolation protocols and then store the strains obtained. In this way they could assess the microbial diversity of different environments. They were able to obtain pure strains. Microbial Sources Selective Media Pure Strain Fermentation Sample treatment aux Strain Collection Extract Library
Since they introduced this method, they started to create a strain collection to keep all the samples and all the strains discovered. They also collected the extract inside a library (extract bank) in a microwell format, so they could be screened automatically. Having a strain collection and the respective extract library is very useful because if you find that one extract is particularly active you can go back to the strain, start fermentation and plan scaling-up to produce and further analyze the molecule. The extract libraries should be compatible with different assays and of good quality. Through this system all the antibiotics known today were discovered during the Golden Age (1940-1950) from soil actinomycetes and fungi. Most of the molecules known are anti-bacterial. All the molecules are classified based on:
- Chemical structure > e.g., aminoglycosides, cephalosporins, penicillins, tetracyclines Mechanism of action > usually it's the inhibition of some macromolecular structure synthesis (like peptidoglycan) Microbial producers Antimicrobial spectrum > the type of microorganisms the molecule is active on Cellular target > it is fundamental to know so that I can use different classes if the microorganism develops resistance to that specific molecule, if the molecules have different targets then there's no risk of cross-resistance
Roughly 30.000 microbial specialized metabolites possessing some kind of activity in vitro are known, most of them are anti-bacterial but also antifungal, antitumoral, algicides, antiparasitic, antivirals, siderophores5 etc. Having seen the success of this method the same process was implemented to find molecules with other kinds of activities, such as: anticancer, immunomodulators, anti-inflammatory, anti- viral, insecticide and antiparasitic compounds. Today we have a different way to classify assay systems:
- Cell-based: where the molecules are tested against a living cell line or microbial culture
- Cell-free: the components of the assays are all extracts (like receptor, enzymes ... ) usually used to study an enzymatic activity or the inhibition of a receptor
In vivo: both in animals and insects, there may be ethical problems and restrictions
DRUG DISCOVERY IN THE INDUSTRY
The advent of industrial companies changed the way we do drug discovery, they started to have private strain collections and to prepare huge extract banks. The process is still biologically guided.
- Analyze the strain collection/extract banks with assays and targets
- Hit > when you select an active extract against a specific target To go from the hit to the lead you need to grow the specific strains, produce the molecule, boost the production and purify it It's important to have a huge amount to be able to perform all the chemical tests needed to profile the molecule
- Lead -> a molecule that has a confirmed biological activity and has been profiled from a chemical point of view
- Optimized lead -> you have to understand the different reactions it has in vivo and if needed you can change them by chemically modifying it
- Clinical candidate > could be the 'natural' molecule or the optimized version