Blotting Techniques for Protein and Nucleic Acid Identification
Slides from University about Blotting Techniques for Protein and Nucleic Acid Identification. The Pdf, a presentation for university-level Biology students, details the general procedure for blotting, focusing on Western Blotting steps and chemiluminescent detection methods.
See more18 Pages

Unlock the full PDF for free
Sign up to get full access to the document and start transforming it with AI.
Preview
Blotting Techniques Overview
"Blotting" is used for the identification of proteins and nucleic acids and is widely used for research and diagnostic purposes. This technique immobilizes the molecule of interest on a support, which is a nitrocellulosic membrane or nylon. It uses hybridization techniques for the identification of the specific nucleic acids or proteins of interest.
The blotting technique is a tool used in the identification of biomolecules such ad DNA, mRNA and protein during different stages of gene expression. Subtypes of blotting such as northern, western & Southern depend upon the target molecule that is being sought.
When a DNA sequence is the foundation or code for a protein molecule, the particular DNA molecule of interest can be blotted using Southern Blotting technique. During gene expression, when the DNA is expressed as mRNA for a protein production, this process can be identified by Northern blotting. Finally, the coded mRNA produces the concerned protein, this protein identification can be done by Western Blotting
General Procedure for Blotting
- General Procedure for blotting
- Homogenize the sample.
- Separation of the molecule of interest by an electrophoresis membrane.
- Transferring the molecules to a nitro cellulosic membrane/ nylon membrane.
- Hybridization or identification of the moleculewestern blotting (proteins)
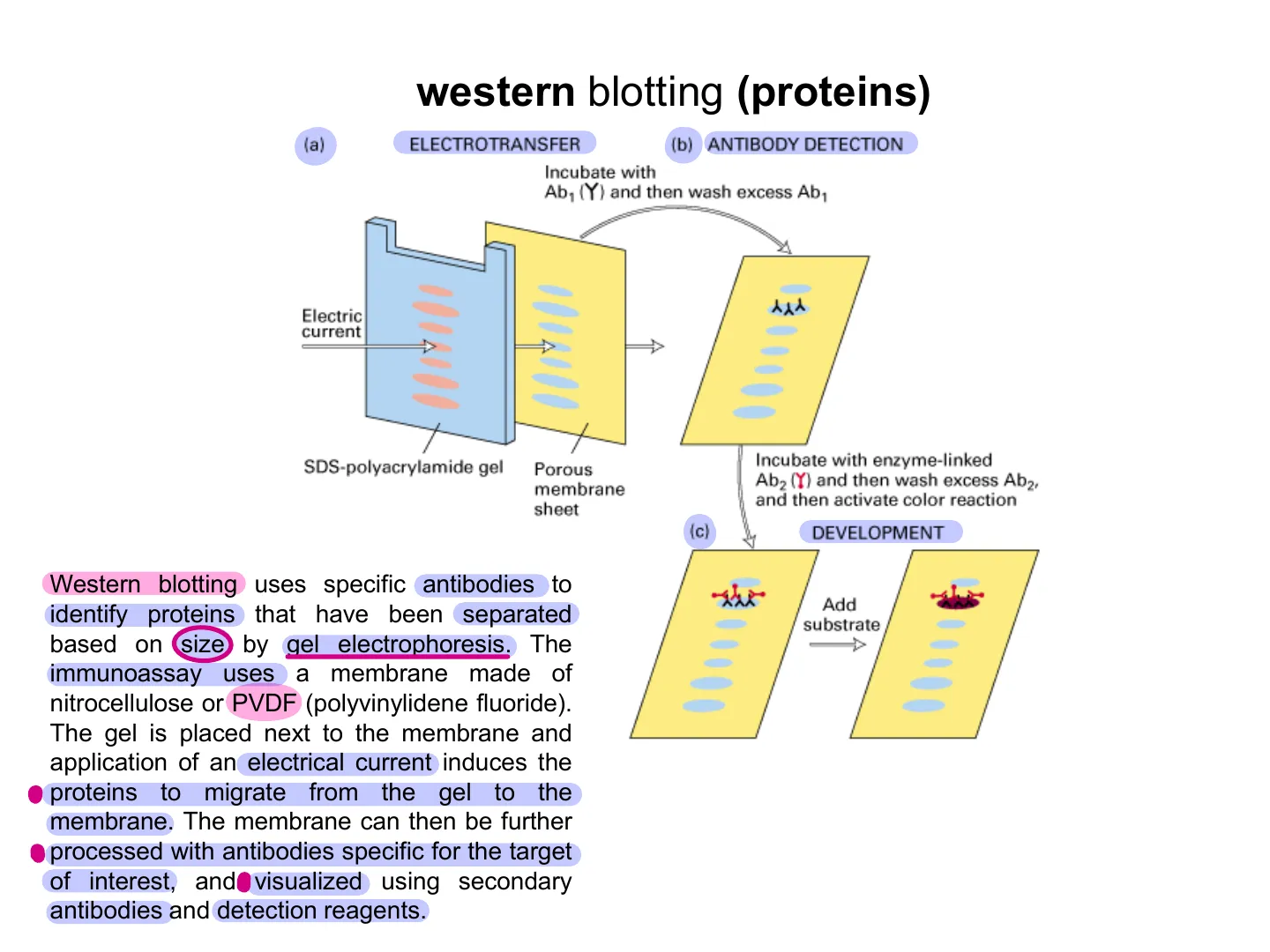
Western Blotting Steps
(a) ELECTROTRANSFER (b) ANTIBODY DETECTION Incubate with Ab1 (Y) and then wash excess Ab1 Electric current WESTERN BLOT STEPS: a) electrotransfer b) antibody detection (Immunoglobulin G: IgG is the most common type of antibody in your blood and other body fluids) c) development SDS-polyacrylamide gel Porous membrane sheet Incubate with enzyme-linked Ab2 (Y) and then wash excess Ab2, and then activate color reaction {c) DEVELOPMENT Add substrate
Western Blot Definition
WESTERN BLOT DEFINITION Western blotting uses specific antibodies to identify proteins that have been separated based on size by gel electrophoresis. The immunoassay uses a membrane made of nitrocellulose or PVDF (polyvinylidene fluoride). The gel is placed next to the membrane and application of an electrical current induces the . proteins to migrate from the gel to the membrane. The membrane can then be further · processed with antibodies specific for the target of interest, and visualized using secondary antibodies and detection reagents.WESTERN BLOT SETUP
Western Blot Setup Components
Western Blot Setup ℮ Cathode + Anode Gel cast between glass plates. Notches cast in top of gel to receive samples. Gel Separated Components · Transfer Membrane Filter Paper Tracking Dye Pads ! Support Grid BufferWESTERN BLOT DETECTION:
Western Blot Detection Signal
- membrane containing transferred protein
- target protein on the membrane
- primary antibody bound to protein
- enzyme-conjugated secondary antibody (bound to primary antibody) transforms the enzyme substrate into a DETECTION SIGNAL (which can be colorimetric or cheluminescent)
Detection Signal (colorimetric or chemiluminescent) Enzyme-conjugate| Secondary Antibody - Enzyme Substrate Primary Antibody Target Protein Membrane Containing Transferred Protein
Western Blot Aim and Method
WESTERN BLOT AIM & METHOD Western blot aims to identify specific proteins within a complex mixture The western blot technique requires samples to be resolved based on size through sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS PAGE), following which they are transferred to and immobilized on a membrane before antibody-based detection.WESTERN BLOT DETECTION:
Western Blot Detection Components
- proteins transferred onto nitrocellulose (human proteins)
- non-cross-reacting blocking protein (e.g. bovine serum albumin)
- enzyme-linked (anti-mouse IgG rabbit polyclonal)
- protein of interest
- primary antibody (anti-human target protein mouse monoclonal)
Enzymes accelerate chemical reactions by decreasing the activation energy ST A energy- EST T S 1 B ES P EP progress of reaction activation energy for catalyzed reaction Substrate Product ... Primary antibody (anti-human target protein mouse monoclonal) Protein of interest m Enzyme-linked (anti-mouse IgG rabbit polyclonal ) ? . Non-cross-reacting blocking protein (e.g. bovine serum albumin) Proteins transferred onto nitrocellulose (human proteins) activation energy for uncatalyzed reaction Enzyme fuctionalizationSDS-PAGE vs Western Blotting
SDS-PAGE vs Western Blotting Comparison
SDS-PAGE western blotting - 97.4 - - 66.2 - - 45.0 - - 31.0 - - 21.5 - -14.4 - SDS gel Immunoblot In the end you got the light emission detected.WESTERN BLOT TYPES:
Western Blot Types
- colorimetric
- cheluminescent
- fluorescent
Colorimetric Chemiluminescence Fluorescence Substrate Substrate Light Product Product * LightColorimetric western blot detection. The left panel demonstrates indirect detection while the right panel shows direct detection.
Colorimetric Western Blot Detection
Indirect and Direct Detection
Indirect detection enzyme Substrate E Primary antibody Detectable product Ag Membrane Direct detection 1. COLORIMETRIC WESTERN BLOT: · indirect detection · direct detection Substrate E Primary antibody Detectable product Ag Membrane Si BASA
Colorimetric Substrate Properties
Colorimetric detection relies on the generation of a colored product that becomes deposited on the western blot, following the conversion of a chromogenic substrate by an appropriate enzyme. The limited sensitivity of chromogenic substrates can make it difficult to optimize them for detecting proteins of low abundance. Colorimetric substrates are suitable for the detection of abundant proteins since the reaction can be monitored visually and allowed to progress until there is adequate color development before being stopped. No specialized equipment is required for visualization of the colored precipitate, and the produced signal is highly stable.ENZYMES USED IN ENZYME-LINKED ANTIBODIES: 1. BCIP 2. HRP
Enzymes in Enzyme-Linked Antibodies
The enzyme used in enzyme-linked antibodies is usually either alkaline phosphatase, which converts colourless 5-bromo-4-chloro- indolylphosphate (BCIP) substrate into a blue product, or horseradish peroxidase (HRP), which, with H2O2 as a substrate, oxidises either 3-amino-9-ethylcarbazole into an insoluble brown product, or 4-chloro-1-naphthol into an insoluble blue product. NEWMAN'S PRODUCI BEST SERO OF AUSTRALIA PREPARED HORSERADISH From an old English recipe ORIGINAL RED LABEY TURN THIS WAY TO OPEN. 250g ml SAB WASABI ORIGINAL S&B WASABI SAUCE He Added Colour mi ZUCCATO dal 1868 Kren Meerrettich Horseradish evet 200g ℮WESTERN BLOT EXAMPLE:
Western Blot Example
Marker 10 ug 1 ug 0.1 ug 0.01 ug Immunoblot analysis marker (lane 1), 10 µg (lane 2), 1 µg (lane 3), 0.1 µg (lane 4), 0.01 ug (lane 5) of BSA samples were resolved by electrophoresis, blotted to nitrocellulose, and probed with Anti-BSA. Proteins were visualized with at anti-rabbit secondary antibody conjugated to HRP and DAB substrate H " H Arrow indicates BSA (69 kDa). ?Chemiluminescent western blot detection.
Chemiluminescent Western Blot Detection
2. CHELUMINESCENT WESTERN BLOT Substrate HRP Light Primary antibody Oxidized products Blocker Ag Ag Membrane Chemiluminescence occurs when a substrate is catalyzed by an enzyme and produces light (photons) as a byproduct of the reaction. The limiting reagent in the reaction is the substrate - as this is exhausted, the light production decreases and eventually stops. However, a well-optimized procedure should produce a stable light output for several hours, allowing consistent and sensitive protein detection.2. CHELUMINESCENT WESTERN BLOT
Enhanced Chemiluminescence (ECL)
- alternative approach = ECL
Light Enhancer O O 2 H2O2 2 H2O NH HO Peroxidase 20- NH OH NH2 ŃH2 N2 Luminol Aminophthalic acid An alternative approach to the detection of horseradish peroxidase is to use the method of enhanced chemiluminescence (ECL). In the presence of hydrogen peroxide and the chemiluminescent substrate luminol horseradish peroxidase oxidises the luminol with concomitant production of light, the intensity of which is increased 1000-fold by the presence of a chemical enhancer. The light emission can be detected by exposing the blot to a photographic film. Corresponding ECL substrates are available for use with alkaline-phosphatase-labelled antibodies.The old-fashion procedure to expose your filter ...
Film Exposure Procedure
X-ray film blot 1 Exposure cassette stainless steel with metal crossbar closureYou needed a darkroom to develop your own film ...
Darkroom Film Development
n n developer stop bath fixer washADVANTAGES & DRAWBACK OF CHELUMINESCENT WESTERN BLOTTING
Chemiluminescent Western Blotting Advantages and Drawbacks
- Advantage: Exposing a chemilumiscent blot to a photographic film increases the threshold for the signal
- Drawback: but still affords a very limited dynamic interval for comparing samples
Soglia Exposing a chemilumiscent blot to a photographic film increases the threshold for the signal, but still affords a very limited dynamic interval for comparing samples. By the way, what would one expect doubling the exposure time? What if exposure time is halved?
Signal-to-Noise Ratio
divido IM 2 Signal-to-noise ratio 10 sec exposure 180.0 160.0 140.0 Signal-to-noise ratio 120.0 100.0 80- 60 40 30 29 28 28 27 27 26 24 23 26 22 19 19 14 20 0- 30 20 15 10 7.5 5.0 3.8 2.5 1.9 1.3 0.63 0.31 0.15 0.08 Sample load, ng X-ray film Can one improve the resolving power of the assay?BENEFITS OF DIGITAL IMAGING OVER FILM
Benefits of Digital Imaging Over Film
Digital imaging systems provide many benefits over film: · Wide dynamic range = 4 logs or better; · Quantitative imaging; · Digital documentation; · Environmentally friendly - no chemicals; • No costly consumables (film and developing); DIGITAL vs FILM Western Blot Images
Digital vs Film Western Blot Images Comparison
Broad dynamic range and superior signal-to-noise ratioDigital vs. Film Western Blot Images Film 4 5 6 Digital 160 143.4 · film Band intensity/lane 7 background 140 O digital 120 100 82.8 80 60 4 5 6 41.0 40 22.5 20.0 20.4 20 15.9 9.4 6.7 3.2 4.6 2.0 2.0 1.4 0.8 0 Lane 1 Lane 2 Lane 3 Lane 4 Lane 5 Lane 6 Lane 7 Lane 8 Broad dynamic range and superior signal-to-noise ampio (WIDE) Signal-to-noise-ratio 11.7Multifluorescence-based "multiplex" analysis
Fluorescent Western Blot: Multiplex Analysis
3. FLUORESCENT WESTERN BLOT: allows Multiplex Analysis (= Digital Imaging) 649-Labeled Secondary Antibody 488-Labeled Secondary Antibody 800-Labeled Secondary Antibody Fluorescence Fluorescence Fluorescence Conjugated Secondary Antibody Conjugated Secondary Antibody Conjugated Secondary Antibody Primary Antibody 1 Primary Antibody 2 Primary Antibody 3 Membrane Schematic of Multiplex Fluorescent Western Blotting