Food processing: reologia e applicazioni nelle scienze alimentari
Documento di Università su Food processing. Il Pdf esplora la reologia, la scienza del flusso e della deformazione dei materiali, con un'enfasi sulle sue applicazioni nel settore alimentare. Questo Pdf di Scienze, utile per l'università, include definizioni, schemi e una cronologia storica della reologia, fornendo una comprensione chiara dei principi reologici e della loro rilevanza pratica.
Mostra di più25 pagine

Visualizza gratis il Pdf completo
Registrati per accedere all’intero documento e trasformarlo con l’AI.
Anteprima
Esame Finale e Lavoro di Gruppo
P. Dordoni Food processing The student will be required to take a final oral exam consisting of 2-3 theoretical questions and in the discussion of the working group. The latter will be carried out during the semester, with an illustration of the topics and aims of the work, and the requirements of the final report (PowerPoint presentation). The work groups may consist indicatively of a maximum of 4-5 students, and the contribution and role of each member must be explicitly indicated in the final report. The final report will be assessed with a mark out of 30. The final mark will be taken as a weighted arithmetic mean of the mark obtained in the oral test (with a 2/3 weight) and that obtained in the group work assessment (with a 1/3 weight).
Definizione di Reologia
Rheology Definition -> "Rheology is the study of the flow of materials that behave in an interesting or unusual manner. Oil and water flow in familiar, normal ways, whereas mayonnaise, peanut butter, chocolate, bread dough, and silly putty flow in complex and unusual ways. In rheology, we study the flows of unusual materials." " ... all normal or Newtonian fluids (air, water, oil, honey) follow the same scientific laws. On the other hand, there are also fluids that do not follow the Newtonian flow laws. These non-Newtonian fluids, for example mayo, paint, molten plastics, foams, clays, and many other fluids, behave in a wide variety of ways. The science of studying these types of unusual materials is called rheology"
Applicazioni Quotidiane della Reologia
We deal with rheology every day. The sensations experienced when tasting creams, squeezing toothpaste, or rubbing skin with body creams could be described by rheology. Rheology considers the fluid and structural properties of raw materials, intermediates, ingredients and final products in the food, pharmaceutical and cosmetic industries. Consumers use subjective tests to evaluate the perceived fluidity and structure of a food product. The purpose of rheology is therefore to make such perceptions objective. Example: ketchup.
Comportamento Reologico dei Prodotti Alimentari
Food products must be formulated to exhibit desired rheological behavior (e.g., ease of pouring from the bottle, controlled flow, stabilization of structure after pouring). Rheology aims to measure the properties of food materials that control their deformation and flow when subjected to external forces (e.g., sucking, pouring, scooping, etc.).
Esempi di Applicazioni della Reologia
Food handling Cream spreadability Spillage of a product from the packaging Food processing Food acceptability Product handling in the plants Keeping the filling in a praline Rating / sensory perception of a product Effort required for chewing, swallowing, etc. Shelf life Staling of bakery products Phase separation in creams Raw materials Processing Consumption RHEOLOGY Development of new products and / or new formulations Design and / or modification of production parameters Characterization of finished products 1P. Dordoni
Importanza della Reologia per i Tecnologi Alimentari
Food processing For food technologists, knowledge of the basics of food rheology is critically important to better understand how process variables affect specific textural characteristics, such as pourability and mouthfeel. Mathematical modeling of rheological behavior makes it possible to predict the performance of food products during exposure to certain processing or experimental conditions. Rheological data should help food technologists and plant engineers design more efficient and economical processes.
Applicazioni nell'Industria Alimentare
In the food industry, applications are: · Process engineering calculations (piping, pumps, extruders, mixers, coatings, heat exchangers, homogenizers, viscometers); · Intermediate or final product quality control; · Shelf life testing; · Evaluation of food consistency by correlation with sensory data; · Analysis of rheological equations of state or constitutive equations -> constitutive equations are used to evaluate food properties at the macroscopic level, but are directly influenced by changes that occur at the microscopic level (molecular behavior). Any equation is just a model of physical reality.
Complessità dei Sistemi Alimentari
Foods are complex systems due to the presence of multiple components whose interactions can drastically affect their rheological behavior.
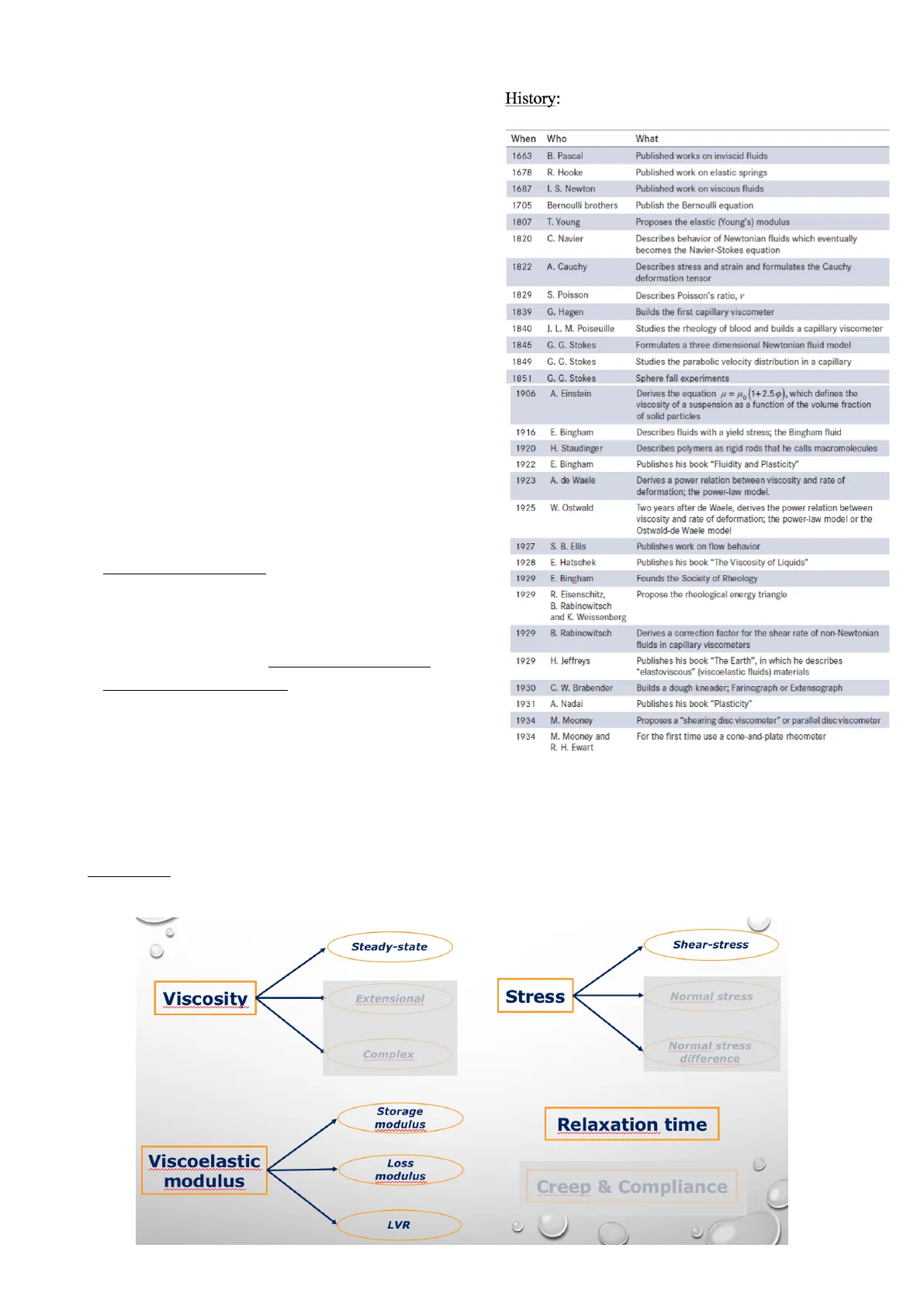
Storia della Reologia
Pionieri e Scoperte
History: When Who What
- B. Pascal Published works on inviscid fluids
- R. Hooke Published work on elastic springs
- I. S. Newton Published work on viscous fluids
- Bernoulli brothers Publish the Bernoulli equation
- T. Young Proposes the elastic (Young's) modulus
- C. Navier Describes behavior of Newtonian fluids which eventually becomes the Navier-Stokes equation
- A. Cauchy Describes stress and strain and formulates the Cauchy deformation tensor
- S. Poisson Describes Poisson's ratio, v
- G. Hagen Builds the first capillary viscometer
- J. L. M. Poiseuille Studies the rheology of blood and builds a capillary viscometer
- G. G. Stokes Formulates a three dimensional Newtonian fluid model
- G. G. Stokes Studies the parabolic velocity distribution in a capillary
- G. G. Stokes Sphere fall experiments
- A. Einstein Derives the equation u= "0(1+2.5¢), which defines the viscosity of a suspension as a function of the volume fraction of solid particles
- E. Bingham Describes fluids with a yield stress; the Bingham fluid
- H. Staudinger Describes polymers as rigid rods that he calls macromolecules
- E. Bingham Publishes his book "Fluidity and Plasticity"
- A. de Waele Derives a power relation between viscosity and rate of deformation; the power-law model.
- W. Ostwald Two years after de Waele, derives the power relation between viscosity and rate of deformation; the power-law model or the Ostwald-de Waele model
- S. B. Ellis Publishes work on flow behavior
- E. Hatschek Publishes his book "The Viscosity of Liquids"
- E. Bingham Founds the Society of Rheology
- R. Eisenschitz, Propose the rheological energy triangle B. Rabinowitsch and K. Weissenberg
- B. Rabinowitsch Derives a correction factor for the shear rate of non-Newtonian fluids in capillary viscometers
- H. Jeffreys Publishes his book "The Earth", in which he describes "elastoviscous" (viscoelastic fluids) materials
- C. W. Brabender Builds a dough kneader; Farinograph or Extensograph
- A. Nadai Publishes his book "Plasticity"
- M. Mooney Proposes a "shearing disc viscometer" or parallel disc viscometer
- M. Mooney and R. H. Ewart For the first time use a cone-and-plate rheometer
Interazioni a Livello Microscopico
What happens within a microscopic volume of our food product when it is subjected to an external stress source? Inter/intramolecular interactions occur; spatial reorganization/rearrangement; stretching/folding of molecules; collapsing of intermolecular bonds.
Parametri Reologici
Parameters: Steady-state Shear-stress Viscosity Extensional Stress Normal stress Complex Normal stress difference Storage modulus Relaxation time Viscoelastic modulus Loss modulus Creep & Compliance LVR 2P. Dordoni
Viscosità
Definizione e Legge di Newton
Il is viscosity! Food processing Viscosity Some fluids flow more easily than others (example glue, honey, oil, water). The tendency of a fluid to flow with ease or with difficulty has been a topic of great practical and intellectual importance to mankind for centuries. One of the first scientists to study this was Newton. Viscosity is anything but the tendency of a fluid to resist to flow. Viscosity is direct connect with speed (velocity). Newton's law -> the resistance which arises from the lack of slipperiness of the parts of the liquid, other things being equal, is proportional to the velocity with which the parts of the liquid are separated from one another. For example water is faster than oil. Lack of slipperiness -> viscosity. Newton's law of viscosity describes the behavior of fluids when they flow. Simply put, viscosity is the "resistance" of a fluid to flow, like a kind of internal friction between its particles. Newton said that: 1. In a fluid, if two layers move over each other at different speeds, there will be a frictional force between them. 2. This force depends on three things: - How fast the two layers are moving relative to each other (the relative speed). - How viscous the substance is (the more viscous it is, the harder it is for the layers to slide over each other, like honey compared to water). - The distance between the two layers. Imagine honey and water. Honey is much more viscous than water, so its internal layers offer more resistance when flowing. Water, being less viscous, flows more easily. Newton observed that in many fluids (called "Newtonian fluids"), this relationship is linear: if you increase the speed or the viscosity, you proportionally increase the force needed to make the fluid flow.
Esperimento delle Piastre Parallele
A = W · L Pair of parallel plates (each of them of area A), separated by a gap (Y) Fluid present in the gap System initially at rest y İY What does happen if we set the upper plate in motion? X V0 V0 = V1 V2 this is kinematic viscosity. p is density. kg 2 - 8 - A NO-SLIP CONDITION V F 0 Y kg kg 102cm kg = 0.1 Pa · s g m Another common unit of measurement adopted for viscosity is the «so-called» Poise [P] 3P. Dordoni
Viscosità di Diverse Sostanze
Different viscosity of substances Food processing Substance Viscosity (cP or mPa.s) Air 1.86 × 10-4 Water (0℃) 1.7921 Water (20℃) 1.000 Water (100℃) 0.2838 20% Sucrose solution (20℃) 1.967 40% Sucrose solution (20℃) 6.223 60% Sucrose solution (20℃) 56.7 80% Sucrose solution (20℃) 40,000 Diethyl ether (20℃) 0.23 Glycerol (20℃) 1759
Influenza della Temperatura sulla Viscosità
Legge di Arrhenius
Temperature and composition of our food change the viscosity. For example water at different temperatures: the interaction of molecules increase. The viscosity of liquids decreases with increasing the temperature by approximately following the Arrhenius law. Arrhenius law -> the law describes how the viscosity of a liquid changes with temperature. Generally, viscosity decreases as temperature increases, and the Arrhenius law can be used to model this relationship. The relationship between viscosity and temperature can be expressed by the equation. Activation energy (Ea) -> viscosity depends on the activation energy required for the molecules of a liquid to flow past each other. At lower temperatures, molecules have less kinetic energy, so more effort is needed for movement, which translates into higher viscosity. The activation energy Ea represents the energy barrier that must be overcome to enable this movement. Effect of temperature (T) -> according to the Arrhenius law, when temperature increases, viscosity decreases exponentially. This happens because as temperature increases, the average kinetic energy of the molecules increases, allowing them to more easily overcome the intermolecular forces that impede flow, thus reducing the fluid's viscosity. Exponential behavior -> the equation shows that the behavior of viscosity as a function of temperature follows an inverse exponential trend. As temperature increases, the exponential 2.0 term decreases, causing viscosity to decrease as well. 1.0 The Arrhenius law for viscosity is particularly useful in fluids such as oils, organic liquids, and polymers, where temperature 0.5 significantly affects viscous behavior. In chemical engineering and materials science, knowing how viscosity changes with 0.2 60 0 20 40 80 temperature is important for designing fluid transport Temperature ("℃) processes, mixing, and flow in pipelines. In summary, the Arrhenius law applied to viscosity describes the exponential dependence of a liquid's viscosity on temperature, related to the energy required to enable molecular movement within the fluid. An opposite behavior is found for gases. Temperature increments cause a greater probability of molecules interactions which, hence, strengthen flow resistance. n is the viscosity of the fluid (often expressed in units such as Pascal- seconds, Pa·s); no is a proportionality constant representing the viscosity of the fluid at a reference temperature; Ea is the activation energy for viscous flow (expressed in Joules/mol); R is the universal gas constant, equal to 8.314 J/(mol·K); T is the absolute temperature in Kelvin.
Viscosità di Soluzioni di Saccarosio
100 | 50 Viscosity (mPa.s) 20 75% Sucrose 10 70% Sucrose 5.0 60% Sucrose 40% Sucrose 20% Sucrose Water L 100 M = Mool E RT 4